New technique maps cancer drug uptake inside living cells
A new analytical method could improve how cancer treatments are designed – by allowing scientists to track, for the first time, exactly where inside a living cell a drug accumulates.
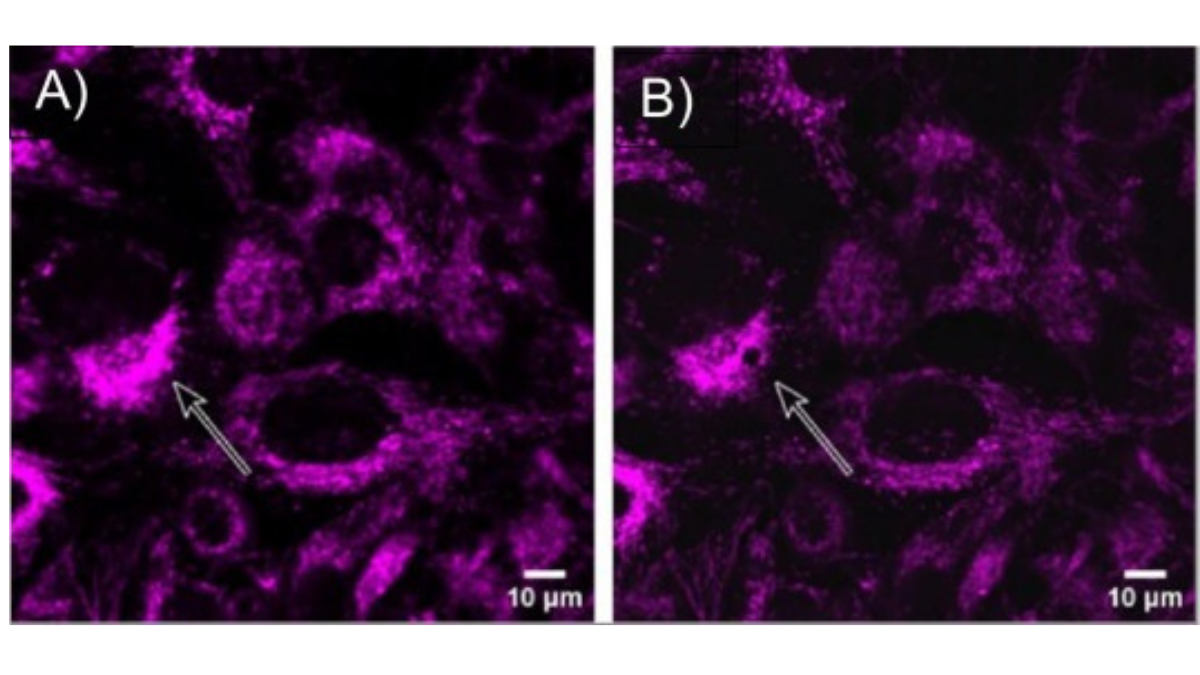
Fluorescently dyed mitochondrial material shown: A) before and B) after sampling, demonstrating the capabilities of the Yokogawa SS2000 to extract subcellular components.
Researchers from the University of Surrey and King’s College London developed the method, which detects trace amounts of metal inside individual living cells and their internal compartments without the need to kill the cells first.
Published in Spectrochimica Acta Part B, the study looked at a class of cancer therapy called targeted radionuclide therapy. This works by attaching a radioactive particle to a molecule that seeks out tumour cells, delivering radiation directly to the cancer. Where inside the cell the drug ends up is critical. A drug that reaches the nucleus causes damage to cancer by targeting DNA. Until now, there was no reliable way to measure this in living cells.
The team used tiny glass capillary tips – ten micrometres wide for whole cells, three micrometres for subcellular structures – to extract individual living pancreatic cancer cells and material from within them, including the powerhouse of cells-mitochondria, under a microscope.
The SEISMIC facility at King’s, a Biotechnology and Biological Sciences Research Council-funded specialist system for extracting material from single living cells, provided the sampling capability. Surrey’s laser ablation inductively coupled plasma mass spectrometry (ICP-MS) facility then enabled detection and measurement of thallium present using LA-ICP-MS – a technique that uses a laser to vaporise minute quantities of material before a mass spectrometer identifies and quantifies the metals within. The combination of capillary sampling at the sub-cellular level and LA-ICP-MS has not been performed before.
The researchers used thallium chloride as a chemically stable stand-in for thallium-201, a radioactive isotope under investigation as a cancer treatment candidate. Thallium was successfully detected in individual cancer cells and, for the first time, inside mitochondria-enriched material extracted from those cells, at extremely low amounts.
Thallium-201 is exciting as a potential cancer therapy precisely because its radiation acts over such a short distance – which means it could destroy tumour cells while sparing the healthy tissue around them. But that precision cuts both ways: the drug has to end up in the right part of the cell to do its job. This method gives us, for the first time, a way to find that out in living cells, and that is a significant step towards making this type of therapy work in practice.Dr Claire Davison from King’s College London
The technique could be extended beyond cancer research to study how any metal-based drug or toxic substance distributes inside living cells. The team identify extracting additional cellular compartments – including the nucleus, where radiation damage to DNA occurs – as a key next step. Improving methods to verify the purity of the extracted subcellular material is also identified as a priority for future development.
The research was supported by the Engineering and Physical Sciences Research Council, the Biotechnology and Biological Sciences Research Council and the Natural Environment Research Council.
Featured Academics
Media Contacts
External Communications and PR team
Phone: +44 (0)1483 684380 / 688914 / 684378
Email: mediarelations@surrey.ac.uk
Out of hours: +44 (0)7773 479911


