Sugar-processing enzyme has a hidden second job – controlling when cells divide
A metabolic enzyme studied for over seven decades has a hidden second function – it can unwind RNA and promote cell cycle progression, an additional function beyond its role in energy production, according to a new study led by the University of Surrey.
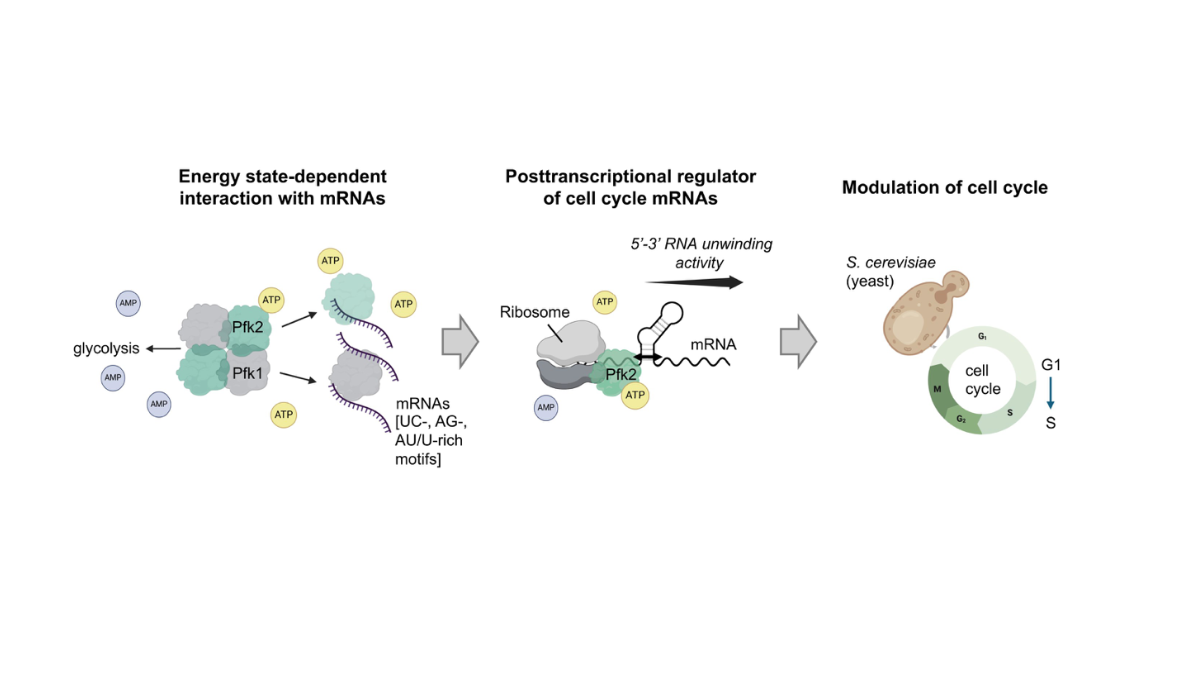
Phosphofructokinase (PFK) is the ‘gatekeeper’ of glycolysis, the ancient and evolutionarily conserved metabolic pathway that breaks down sugar to generate energy. In the yeast Saccharomyces cerevisiae, PFK is made up of two subunits – Pfk1 (α) and Pfk2 (β). While both have long been understood as metabolic partners, the Surrey-led team has discovered that Pfk2 possesses an entirely separate capability. It binds hundreds of messenger RNAs (mRNA) inside cells, unwinds short double-stranded RNA in a specific direction, and actively promotes the translation of genes that drive cell division.
Published in Nucleic Acids Research, the study shows that without Pfk2, yeast cells grow more slowly, become significantly larger and struggle to progress from the G1 to S phase of the cell cycle – a critical transition point where cells commit to division. Crucially, reintroducing a version of Pfk2 that cannot perform glycolysis still rescued these defects, confirming that the enzyme’s role in cell division is independent of its metabolic function.
The research team used a combination of RNA sequencing, biochemical assays (laboratory tests to study molecular behaviour) and proteomics (large-scale analysis of proteins) to build their case. They identified over 800 mRNAs that Pfk2 binds in living cells, many of which code for proteins involved in controlling the mitotic cell cycle (the process by which a cell divides into two). Using tests that use light signals to track RNA strands being pulled apart in real time, the research team showed that Pfk2 – but not Pfk1 – can unwind short double-stranded RNA molecules with a specific directionality, a function normally associated with dedicated RNA helicase enzymes (specialised proteins whose primary job is to unwind RNA).
Polysome profiling (a technique that separates cell contents to reveal which mRNAs are actively being made into proteins) revealed that in cells lacking Pfk2, mRNAs for critical cell cycle regulators – including the G1 cyclin CLN3 (a protein that triggers the start of cell division) and the spindle checkpoint protein BUB3 (a protein that ensures chromosomes are correctly separated) – shifted dramatically away from ribosomes, indicating they were no longer being efficiently translated into proteins. Proteomics confirmed reduced levels of cell cycle proteins in Pfk2 deletion mutants (cells where the gene coding for Pfk is removed).
The team proposes a “molecular relay switch” model. When cellular energy is low, PFK adopts its enzymatically active state and focuses on glycolysis. When energy is abundant, Pfk2 shifts to a low-activity shape that enhances its ability to bind and unwind RNA, promoting the translation (production of proteins from RNA instructions) of cell cycle genes and enabling cell division. This creates a direct molecular link between a cell’s metabolic state and its decision to proliferate.
The research was funded by the Biotechnology and Biological Sciences Research Council (BBSRC), Cancer Research UK and the Engineering and Physical Sciences Research Council (EPSRC). International collaborators included teams at the Cancer Research UK Scotland Institute, the University of Osnabrück, the University of Basel and Ulm University.
[ENDS]
Notes to editors
- Professor André Gerber is available for interview; please contact mediarelations@surrey.ac.uk to arrange.
- The full paper, “The yeast phosphofructokinase β-subunit has RNA unwinding activity and modulates cell cycle progression”, is published in Nucleic Acids Research.
- Images are available upon request.
- For more information, please contact the University of Surrey’s press office via mediarelations@surrey.ac.uk
Media Contacts
External Communications and PR team
Phone: +44 (0)1483 684380 / 688914 / 684378
Email: mediarelations@surrey.ac.uk
Out of hours: +44 (0)7773 479911

