Professor Derk-Jan Dijk
Academic and research departments
School of Biosciences, Surrey Sleep Research Centre, Faculty of Health and Medical Sciences.About
Biography
Derk-Jan Dijk PhD, FRSB, FMedSci, is Professor of Sleep and Physiology, Distinguished Professor at the University of Surrey, Director of the Surrey Sleep Research Centre. He has been a Royal Society-Wolfson Research Merit Award holder, a Senior Research Associate in the Institute of Pharmacology at the University of Zurich, an Assistant Professor of Medicine at Harvard Medical School, and an Associate Neuroscientist in the Brigham and Women's Hospital in Boston.
Dr Dijk has 40 years of experience in clinical sleep research. His current research interests include the circadian and homeostatic regulation of sleep; the contribution of sleep to brain function in healthy ageing and dementia; the role of circadian rhythmicity in sleep regulation; identification of novel-biomarkers for sleep debt status and circadian rhythmicity, susceptibility to the negative effects of sleep loss; understanding age and sex related differences in sleep physiology and sleep disorders. His research has been or is is funded by the Dementia Research Institute, the Biotechnology and Biological Science Research Council, the Wellcome Trust, The Air Force Office of Scientific Research, Philips Lighting and several major pharmaceutical companies.
Dr Dijk has published more than 300 research and review papers in the area of sleep and circadian rhythms. Dr Dijk is invited frequently to speak at international sleep meetings and he has given opening and plenary lectures for the joint meeting of the Canadian Sleep Society, American Academy of Sleep Medicine and Sleep Research Society, The European Sleep Research Society and the Hong Kong Sleep Medicine Society.
Dr Dijk has served as an Associate and Deputy Editor to SLEEP and Editor of the Journal of Sleep Research. He also serves as consultant to the pharmaceutical industry.
Areas of specialism
News
In the media
ResearchResearch interests
Sleep is an important determinant of wellbeing and health. Understanding how sleep is regulated and how it contributes to mental and physical health, cognition and well-being is at at the centre of the research in the Surrey Sleep Research Centre. Using approaches ranging from genetics to behavioural assessments, and with an emphasis on sleep physiology, EEG analysis and circadian rhythms, the functional significance of sleep is investigated across the adult life span.
Research interests
Sleep is an important determinant of wellbeing and health. Understanding how sleep is regulated and how it contributes to mental and physical health, cognition and well-being is at at the centre of the research in the Surrey Sleep Research Centre. Using approaches ranging from genetics to behavioural assessments, and with an emphasis on sleep physiology, EEG analysis and circadian rhythms, the functional significance of sleep is investigated across the adult life span.
Supervision
Postgraduate research supervision
I supervise on the following courses:
Sustainable development goals
My research interests are related to the following:



Publications
INTRODUCTION Research in the field of sleep, aging, and dementia is rapidly growing. Consensus guidance is needed to facilitate high‐quality research, comparability, and consistency. METHODS A modified Delphi consensus study was conducted by the Sleep and Circadian Rhythms Professional Interest Area of the International Society to Advance Alzheimer's Research and Treatment (ISTAART) and an international panel of experts to establish recommendations for future research. RESULTS After two voting rounds, the group (1) determined by consensus the most relevant sleep and circadian features to assess in the context of aging and dementia research, (2) established recommendations on data acquisition and report for future studies, and (3) identified high‐priority future directions. DISCUSSION This expert consensus study provides guidance to develop high‐quality sleep and circadian research in the context of aging and dementia. Similar recommendations will need to be established for clinical practice. Highlights We report the results of an international expert consensus study. Sleep and circadian rhythms features relevant to aging and dementia were identified. Recommendations on data acquisition and reporting were established. High‐priority future research directions were identified.
Introduction We investigated whether the relationship between daytime sleepiness and previous night’s sleep duration changes with age using a protocol in which individuals were studied both after habitual sleep duration and after multiple days with extended sleep opportunities. Methods 35 younger (18-32 years old, 18 F) and 18 older (60-76 years old, 6 F) healthy participants were studied using polysomnography in an in-lab protocol (Klerman and Dijk, Curr. Biol. 2008). During night 1 (SP1), participants were scheduled to sleep based on their at-home sleep schedules. On day 2, the 5 sessions of a MSLT (MSLT_pre) began 2 hours after SP1 ended .Participants then began a schedule with 16 hrs (12 hrs at night and 4 hrs during day) of sleep opportunity in bed per 24 hrs for 7 days, followed by a second set of MSLTs (MSLT-post). Total sleep time (TST) for the nighttime sleep episodes prior to both sets of MSLTs was calculated. The median sleep latency and percent of the MSLT sessions in which participants fell asleep (PctSlept) within MSLT_pre and MSLT_post were calculated. Results Total sleep time on SP1 ranged from 5.4-9.7hr; only Younger participants had a TST >8.0 hr. Therefore, for analyses comparing Younger and Older participants, only TST of < 8.0 hr was used (21 Younger, 18 Older). All participants fell asleep at least once during MSLT_pre. During MSLT_post, 32% of Younger and 8% of Older participants did not fall asleep during any of the MSLT sessions (i.e., PctSlept=0); therefore, latency-based analyses cannot be done for MSLT_post data. There was a negative correlation between SP1 TST and median sleep latency for MSLT_pre for Younger (Pearson R=0.35; p=0.049) but not Older participants. There was a negative correlation between SP1 TST and PctSlept for Younger (Pearson R=-0.47, p=0.006), but not Older participants. There was no relationship between TST before MSLT_post and the PctSlept for either Younger or Older participants. Repeated measures ANOVA testing (MSLT_pre vs MSLT_post) found a time (p< 0.001) and time*agegroup (p=0.008) effect for PctSlept. Conclusion The relationship between the prior night’s sleep and daytime sleepiness may change with age. Support (if any) NIH R01- HL166205, R01-HD107064
This chapter describes how sleep is regulated by a process that tracks sleep–wake history and a circadian process. It focusses on human studies, but it is emphasized that many principles of this twoprocess model for the maintenance of homeostasis also apply to animals. It reviews the empirical fundamentals, conceptual representation, and a few relevant mechanistic details of this twoprocess system. It explains how the separate contributions of sleep–wake history and circadian rhythmicity can be assessed in protocols such as the forced desynchrony and constant routine protocol. It summarizes how sleep timing and structure as well as brain function during wakefulness are modulated by these two processes. It also reviews how environmental factors such as light and social factors such as work schedules influence the timing of sleep and circadian rhythms. Implications for the understanding of sleep timing disorders are discussed.
Bioelectronic implants for neurostimulation aim to steer disordered neurophysiological processes back towards a healthy state. However, physiology is subject to biological rhythms, including the circadian rhythm and the sleep-wake cycle. These predictable rhythms affect disease symptomatology, biomarkers used in closed-loop therapies, and a physiological system's expected response to stimulation. Therefore, therapeutic devices should incorporate feedforward elements to align algorithm parameters with predictable changes in physiological state, as a parallel of physiological rheostatic control. Here we introduce the DyNeuMo-2c, the first clinical-grade implant capable of delivering closed-loop neurostimulation while flexibly changing its functional configuration according to time of day. The device can chronically measure brain activity and motion state to track potential biomarker patterns in natural, out-of-clinic settings, allowing identification and targeting of patient-specific chronotypes. The system implements a hierarchical control flow, with baseline therapy set by a circadian scheduler, and adaptive policies layered to take effect based on specific biomarkers indicating patient and disease state. Using a benchtop validation setup, we demonstrate that the system has the required capabilities for delivering time-contingent closed-loop therapy in two established clinical use cases: Parkinson's disease and epilepsy. Next, we deploy the system to deliver closed-loop deep brain stimulation in a healthy non-human primate model of vigilance, highlighting the importance of synchronisation between device operation and physiological state in various conditions (task performance, unconstrained behaviour, and sleep). Time-of-day-dependent adaptation of closed-loop stimulation enabled modulation of both vigilance and behaviour. Overall, the novel device architecture provides a proof-of-concept for delivering time-contingent therapy in chronic therapeutic settings where biological rhythms are of key importance.
Introduction Cross-sectional and interventional studies have demonstrated that sleep has a significant impact on waking brain function, including alertness and cognitive performance. Few studies have assessed whether spontaneous night-to-night variation in sleep is associated with variation in brain function within an individual. How this compares to inter-individual variation in sleep and cognition and their associations also remains largely unknown. These questions are of particular interest in the context of aging because both sleep and cognitive abilities are altered.Methods Furthermore, older people have been reported to be less sensitive to sleep loss. Here, we investigated the relationship between sleep and cognition by quantifying associations between intra-individual variation in sleep and cognition, along with associations between inter-individual variation in sleep and cognition, in 35 cognitively intact older adults (70.8 +/- 4.9 years; mean +/- SD; 14 female individuals) living in the community. Subjective and actigraphic sleep measures and daily digital assessments of cognition (9 cognitive tests; 19 variables) were obtained over a 2-week period. The cognitive test battery probed a wide range of cognitive functions, including reaction time, working memory, attention, and problem-solving. Principal component analysis (PCA) identified four principal sleep components, namely, sleep duration, sleep efficiency, subjective sleep quality, and nap effect. Mixed model analyses were conducted with mean and deviation-from-the-mean cognitive variables to quantify how inter- and intra-individual variations in sleep were associated with inter- and intra-individual variations in cognition.Results Longer sleep duration was associated with faster reaction times in both the inter- and intra-individual analyses and with reduced errors in the inter-individual analyses. Higher sleep efficiency was associated with faster reaction times in both the intra- and inter-individual analyses. In contrast, aspects of cognition relating to learning, visual memory, verbal reasoning, and verbal fluency were not associated with sleep.Discussion These data show that, in older people, some aspects of waking function are sensitive to night-to-night variation in sleep duration and efficiency, implying that interventions targeting these aspects of sleep may be beneficial for waking function in aging.
Background Changes in sleep patterns are common in Alzheimer’s disease and impact the quality of life of both people living with Alzheimer’s (PLWA) and their caregivers. Longitudinal recordings and assessment of night‐to‐night variations in sleep and physiology can improve our understanding of how sleep influences clinical outcomes and caregiver wellbeing. Method We collected sleep diary and contactless sleep technology data (Withings sleep analyser, WSA) in community dwelling PLWA (N = 16, Age = 72.8 ± 5.7 years, SMMSE = 27.1 ± 1.4), caregivers (N = 8, Age = 74.4 ± 3.8 years, SMMSE = 28.6 ± 0.7) and healthy older adults (N = 23, Age = 70.7 ± 5.5 years, SMMSE = 29.0 ± 0.9) for a period of upto 14 days at home. Group differences were analysed using a mixed model with participants as random effect while coefficients of variation were used to quantify the intra‐individual variability. Result Sleep diary and contactless sleep and heart rate variables were successfully collected for 624 (sleep diary) and 647 (WSA) nights. Analyses of group means of the sleep diary measures revealed that the self‐reported sleep period was significantly longer (p
The alternation of sleep and wakefulness is a major determinant of the structure and quality of life. The sleep–wake cycle is regulated by a fine-tuned balance between two physiological processes: sleep homeostasis, which measures sleep debt, and circadian rhythmicity, which determines the optimal internal (biological) time for sleep and wakefulness. Sleep homeostasis and circadian rhythmicity together influence many aspects of sleep, such as the time it takes to fall asleep, the timing of awakening and the interruptions of sleep, as well as the duration of rapid-eye-movement sleep, slow-wave sleep, and specific brainwaves during sleep, such as sleep spindles. Alterations in the balance between sleep homeostasis and circadian rhythmicity contribute to sleep phenotypes such as morningness–eveningness and short–long sleepers as well as sleep disturbances. Emerging insights into the environmental, behavioral, physiological, neurochemical, and molecular-genetic determinants of sleep homeostasis and circadian rhythmicity provide new avenues for the understanding and improvement of the sleep–wake cycle.
Background Disruption in diurnal rest‐activity rhythms is a hallmark of Alzheimer’s disease. Currently, we know little about how physiology, symptoms, and biomarkers change over the 24‐hour day in people living with Alzheimer’s disease. In particular, we don’t know whether plasma biomarkers of neurodegeneration, which offer promise as diagnostic or stratification tools, vary with time of day, and whether these associate with the circadian markers melatonin and cortisol. Methods We studied 38 participants (70.8 ± 7.6 years; mean ± SD) in a 27‐hour laboratory protocol including a sleep episode with either two blood samples taken 12 hours apart (n = 17) or 3‐hourly blood sampling for 24 hours (n = 21). The participants were people living with mild Alzheimer’s disease (PLWA, n = 8), their partners/caregivers (n = 6), and cognitively intact older adults (n = 24). The neurodegeneration biomarkers phosphorylated tau (p‐tau217), amyloid‐beta 40 (Aβ40), amyloid‐beta 42 (Aβ42), glial fibrillary acidic protein (GFAP), and neurofilament light (NfL) levels were measured with single molecule array technology. A mixed linear model (PROC MIXED, SAS) was run to assess the impact of time‐of‐day, group, and any interaction. The gold‐standard markers of the circadian clock, plasma melatonin and cortisol, were measured using liquid chromatography mass spectrometry (LC‐MS) for the 24‐hour timeseries (n = 21). Correlations were run (R) to explore relationships between 24‐hour levels of dementia biomarkers, melatonin and cortisol. Results Diurnal rhythms in melatonin and cortisol were observed in all participant groups. Significant time of day variation was observed for p‐tau217, Aβ40, Aβ42, NfL, and a significant effect of group for p‐tau217 with highest levels in PLWA. There were no significant correlations between 24‐hour levels of melatonin or cortisol and any of the neurodegeneration biomarkers. Conclusions Time of day variation was observed in central circadian hormone markers and commonly used biomarkers of dementia in all participants. Outputs of the central circadian clock were not markedly altered in PLWA, and there was no relationship between 24‐hour levels of these outputs and blood‐based biomarkers of dementia. Time of day variation in biomarkers may be relevant to their implementation and interpretation in research and clinical practice.
(iScience 27, 109331; March 15, 2024) Due to errors introduced during the drafting of the manuscript, the STAR Methods text under the “rhythmic modelling” heading within the “quantification and statistical analysis” section included incorrect mathematical formulae and references. The authors apologize for this oversight. The STAR Methods text has been corrected in the published article.
•ASL MRI used to assess cerebral perfusion during pharmacologically induced sleep.•Global grey and white matter CBF significantly reduced during sleep vs wake.•Voxelwise analysis revealed cortical and subcortical perfusion decreases.•Exploratory ROI analysis showed reduced flow in ascending arousal nuclei. Cerebral blood flow (CBF) reductions during non-REM sleep have been consistently demonstrated in positron emission tomography (PET) studies, but PET’s limitations restrict its use. Arterial spin labeling (ASL) MRI allows repeated, noninvasive CBF measurement but has rarely been applied to sleep. This study examined global and regional CBF changes during wakefulness and pharmacologically induced sleep after sleep deprivation, with an exploratory focus on the ascending arousal network (AAN). Twenty-one healthy participants (13 male, 8 female; mean age = 22.4 ± 3.1 years) underwent ASL MRI during wake and sleep. Sleep was facilitated with 10 mg Zolpidem following sleep deprivation and verified through reduced button-press responses and increased high-frequency heart rate variability (HF-HRV). Pseudo-continuous ASL was acquired with a labeling duration of 1.9 s and post-labeling delay of 1.8 s. Sleep was confirmed in 19 participants; one was excluded for excessive head displacement, resulting in a final sample of 18. Grey matter CBF decreased from 59.7 ± 9.0 to 49.5 ± 8.1 ml/100g/min (p < 0.0001), and white matter CBF from 28.7 ± 4.9 to 25.1 ± 3.0 ml/100g/min (p < 0.001). Voxelwise analysis revealed reductions in cingulate, medial prefrontal cortex, precuneus, insula, thalamus, and caudate. ROI analysis indicated decreases in AAN regions including the Locus Coeruleus (−29.3 %, p = 0.0019), Medial Reticular Formation (−22.8 %, p = 0.0010), Pontis Oralis (−23.4 %, p = 0.0044), and Pedunculopontine Nucleus (−20.8 %, p = 0.0023). Observed perfusion decreases are consistent with PET evidence of cortical and thalamic downregulation during sleep and indicate suppression of brainstem arousal nuclei. These findings provide a detailed ASL characterization of cortical and brainstem perfusion changes during pharmacologically induced sleep, extending PET work and offering new insight into sleep-related arousal systems.
Our study introduced the 2023 UK Biobank sleep questionnaire and described variation in sleep health dimensions and prevalence of disordered sleep. A questionnaire comprising validated measures and bespoke items was developed to capture key self-reported domains of sleep health and symptoms of sleep disorders. We quantified cohort prevalence of operationally defined sleep disorders and assessed patterning of sleep health dimensions across key sociodemographic and clinically relevant variables. 183,704 individuals completed at least one module of the questionnaire after email invitation (representing 56% of those with an active email address), and an additional 1,352 individuals completed via the participant website. A total of 185,056 individuals were included in the analysis. Respondents were predominately from a White ethnic background (96.8%), had a mean age of 69.9 (SD, 7.5) years, 57.9% were female, and 25.5% were in employment. Compared to non-respondents, respondents were more likely to be female, tended to be better educated, healthier, and exhibit lower levels of socioeconomic deprivation, although baseline sleep variables were similar between respondents and non-respondents. Around 40% of respondents reported sleep duration less than 7 hours and 49% reported poor sleep quality (Pittsburgh Sleep Quality Index > 5). Approximately one-quarter (25.2%) met criteria for at least one operationally defined sleep disorder, with insomnia being the most common (14.4%) followed by obstructive sleep apnoea (8.0%), restless legs syndrome (4.1%), and frequent nightmares (3.7%). Sleep disorders were associated with higher levels of anxiety, depression, fatigue, and cognitive complaints. Poor sleep quality and operationally defined sleep disorders are common in the UK Biobank cohort. Sleep questionnaire data can now be integrated with a range of biomedical information to advance understanding of sleep. Statement of significance A comprehensive sleep questionnaire was introduced to the UK Biobank, with over 185,000 participants providing data. Overall, respondents reported relatively poor sleep quality; 40% reported sleep duration less than 7 hours, and 25% met criteria for at least one sleep disorder. Enhanced assessment of sleep in UK Biobank now enables integration with extensive biomedical data, including genetic, wearable, imaging, lifestyle, biomarker, and electronic health record data, offering opportunities to investigate the biological and environmental factors that influence sleep and circadian systems, and their impact on health.
Sleep is a fundamental part of our lives; yet, how our brain falls asleep remains one of the most enduring mysteries of neuroscience. Here we report a new conceptual framework to analyze and model this phenomenon. The framework represents the changes in brain electroencephalogram activity during the transition into sleep as a trajectory in a normalized feature space. We use the framework to show that the brain's wake-to-sleep transition follows bifurcation dynamics with a distinct tipping point preceded by a critical slowing down. We validate the bifurcation dynamics in two independent datasets, which include more than 1,000 human participants. Finally, we demonstrate the framework's utility by predicting a person's progression into sleep in real time with seconds temporal resolution and over 0.95 average accuracy.
Objective: Ultra-wideband (UWB) radar technology has emerged as a promising alternative for creating portable and cost-effective in-home monitoring devices. Although there exists good evidence supporting its effectiveness in sleep monitoring, previous studies predominantly focus on younger, healthy participants. This research evaluates the applicability of commercial impulse UWB radar for sleep monitoring in older people and people with neurodegenerative disorders (NDDs). Methods: 47 older people (mean age: 71.2 \pm 6.5, 18 with prodromal or mild Alzheimer's disease) participated in our overnight sleep trial with polysomnography (PSG) and UWB radar monitoring. Data processing based on multivariate empirical mode decomposition (MEMD) was employed to reconstruct cardiopulmonary activity and limb movements from radar signals. 29 features were extracted from the radar signals, and sleep stages were classified using a sequence-to-sequence neural network. Additionally, a cross-entropy-based approach was used to quantify uncertainties in the radar classification model and provide confidence in the classification. Results: The UWB radar system demonstrated high accuracy in detecting body movements, reconstructing respiratory patterns, and monitoring heart rate. For sleep stage classification, the results showed a Kappa coefficient of 0.63 and an average accuracy of 74.4% across wake, REM sleep, light sleep (N1 + N2), and deep sleep (N3) categories. Conclusion: The proposed method reliably monitors physiological changes during sleep, which suggests its potential as a cost-effective alternative to traditional sleep monitoring devices. Significance: The findings underscore the viability of UWB radar as a nonintrusive, forward-looking sleep assessment tool that could significantly benefit care for older people and people with neurodegenerative disorders.
This repository contains raw data, sleep scoring, and data to create the figures for the paper: "Closed-loop auditory stimulation targeting alpha and theta oscillations during REM sleep induces phase-dependent power and frequency changes" by Valeria Jaramillo, Henry Hebron, Sara Wong, Giuseppe Atzori, Ullrich Bartsch, Derk-Jan Dijk*, Ines R. Violante* (* contributed equally). Journal article has been published in SLEEP and can be found here: https://doi.org/10.1093/sleep/zsae193 Code can be found here: https://gitlab.surrey.ac.uk/nemo/RSN Please cite as indicated under 'Citation' on this page. More information on the datafiles can be found in the README.
The two-process model (2pm) of sleep regulation is a conceptual framework and consists of mathematical equations. It shares similarities with models for cardiac, respiratory and neuronal rhythms and falls within the wider class of coupled oscillator models. The 2pm is related to neuronal mutual inhibition models of sleep-wake regulation. The mathematical structure of the 2pm, in which the sleep-wake cycle is entrained to the circadian pacemaker, explains sleep patterns in the absence of 24 h time cues, in different species and in early childhood. Extending the 2pm with a process describing the response of the circadian pacemaker to light creates a hierarchical entrainment system with feedback which permits quantitative modelling of the effect of self-selected light on sleep and circadian timing. The extended 2pm provides new interpretations of sleep phenotypes and provides quantitative predictions of effects of sleep and light interventions to support sleep and circadian alignment in individuals, including those with neurodegenerative disorders.
Mechanisms regulating human sleep and physiology have evolved in response to rhythmic variation in environmental variables driven by the Earth’s rotation around its axis and the Sun. To what extent these mechanisms are operable in vulnerable people who are primarily exposed to the indoor environment remains unknown. We analyzed 26 523 days of data from outdoor and indoor environmental sensors and a contactless behavior-and-physiology sensor tracking bed occupancy, heart, and breathing rate in 70 people living with dementia. Indoor light and temperature, sleep timing, duration, and fragmentation, as well as the timing of the heart rate minimum, all varied across seasons. Beyond the effects of season, higher bedroom temperature and less bright indoor daytime light are associated with more disrupted sleep and higher respiratory rate. This sensitivity of sleep and physiology to ecologically relevant variations in indoor environmental variables implies that implementing approaches to control indoor light and temperature can improve sleep.
Biomarkers are valuable tools in a wide range of human health areas including circadian medicine. Valid, low-burden, multivariate molecular approaches to assess circadian phase at scale in people living and working in the real world hold promise for translating basic circadian knowledge to practical applications. However, standards for the development and evaluation of these circadian biomarkers have not yet been established, even though several publications report such biomarkers and claim that the methods are universal. Here, we present a basic exploration of some of the determinants and confounds of blood-based biomarker development for suprachiasmatic nucleus (SCN) phase by reanalysing publicly available data sets. We compare performance of biomarkers based on three feature-selection methods: Partial Least Squares Regression, ZeitZeiger, and Elastic Net, as well as performance of a standard set of clock genes. We explore the effects of training sample size and the impact of the experimental protocols from which training samples are drawn and on which performance is tested. Approaches based on small sample sizes used for training are prone to poor performance due to overfitting. Performance to some extent depends on the feature-selection method, but at least as much on the experimental conditions from which the biomarker training samples were drawn. Performance of biomarkers developed under baseline conditions does not necessarily translate to protocols that mimic real-world scenarios such as shiftwork in which sleep may be restricted or desynchronized from the endogenous circadian SCN phase. The molecular features selected by the various approaches to develop biomarkers for the SCN phase show very little overlap although the processes associated with these features have common themes with response to steroid hormones, that is, cortisol being the most prominent. Overall, the findings indicate that establishment of circadian biomarkers should be guided by established biomarker-development concepts and foundational principles of human circadian biology.
Sleep disorders are a prevalent problem among older adults, yet obtaining an accurate and reliable assessment of sleep quality can be challenging. Traditional polysomnography (PSG) is the gold standard for sleep staging, but is obtrusive, expensive, and requires expert assistance. To this end, we propose a minimally invasive single-channel single ear-EEG automatic sleep staging method for older adults. The method employs features from the frequency, time, and structural complexity domains, which provide a robust classification of sleep stages from a standardised viscoelastic earpiece. Our method is verified on a dataset of older adults and achieves a kappa value of at least 0.61, indicating substantial agreement. This paves the way for a non-invasive, cost-effective, and portable alternative to traditional PSG for sleep staging.
Circadian clocks drive cyclic variations in many aspects of physiology, but some daily variations are evoked by periodic changes in the environment or sleep–wake state and associated behaviors, such as changes in posture, light levels, fasting or eating, rest or activity and social interactions; thus, it is often important to quantify the relative contributions of these factors. Yet, circadian rhythms and these evoked effects cannot be separated under typical 24-h day conditions, because circadian phase and the length of time awake or asleep co-vary. Nathaniel Kleitman’s forced desynchrony (FD) protocol was designed to assess endogenous circadian rhythmicity and to separate circadian from evoked components of daily rhythms in multiple parameters. Under FD protocol conditions, light intensity is kept low to minimize its impact on the circadian pacemaker, and participants have sleep–wake state and associated behaviors scheduled to an imposed non-24-h cycle. The period of this imposed cycle, Τ , is chosen so that the circadian pacemaker cannot entrain to it and therefore continues to oscillate at its intrinsic period ( τ , ~24.15 h), ensuring circadian components are separated from evoked components of daily rhythms. Here we provide detailed instructions and troubleshooting techniques on how to design, implement and analyze the data from an FD protocol. We provide two procedures: one with general guidance for designing an FD study and another with more precise instructions for replicating one of our previous FD studies. We discuss estimating circadian parameters and quantifying the separate contributions of circadian rhythmicity and the sleep–wake cycle, including statistical analysis procedures and an R package for conducting the non-orthogonal spectral analysis method that enables an accurate estimation of period, amplitude and phase. This protocol describes how to design, implement and analyze the data from a forced desynchrony protocol to assess endogenous circadian rhythmicity and to separate circadian from evoked components of daily rhythms in physiology and behavior.
Study Objectives: Portable electroencephalography (EEG) devices offer the potential for accurate quantification of sleep at home but have not been evaluated in relevant populations. We assessed the Dreem headband (DHB) and its automated sleep staging algorithm in 62 older adults [age (mean±SD) 70.5±6.7 years; 12 Alzheimer’s]. Methods: The accuracy of sleep measures, epoch-by-epoch staging, and the quality of EEG signals for quantitative EEG (qEEG) analysis were compared to standard polysomnography (PSG) in a sleep laboratory. Results: The DHB algorithm accurately estimated total sleep time (TST) and sleep efficiency (SEFF) with a Symmetric Mean Absolute Percentage Error (SMAPE)
Background: Contactless sleep technologies (CSTs) hold promise for longitudinal, unobtrusive sleep monitoring in the community and at scale. They may be particularly useful in older populations wherein sleep disturbance, which may be indicative of the deterioration of physical and mental health, is highly prevalent. However, few CSTs have been evaluated in older people. Objective: This study evaluated the performance of 3 CSTs compared to polysomnography (PSG) and actigraphy in an older population. Methods: Overall, 35 older men and women (age: mean 70.8, SD 4.9 y; women: n=14, 40%), several of whom had comorbidities, including sleep apnea, participated in the study. Sleep was recorded simultaneously using a bedside radar (Somnofy [Vital Things]: n=17), 2 undermattress devices (Withings sleep analyzer [WSA; Withings Inc]: n=35; Emfit-QS [Emfit; Emfit Ltd]: n=17), PSG (n=35), and actigraphy (Actiwatch Spectrum [Philips Respironics]: n=18) during the first night in a 10-hour time-in-bed protocol conducted in a sleep laboratory. The devices were evaluated through performance metrics for summary measures and epoch-by-epoch classification. PSG served as the gold standard. Results: The protocol induced mild sleep disturbance with a mean sleep efficiency (SEFF) of 70.9% (SD 10.4%; range 52.27%-92.60%). All 3 CSTs overestimated the total sleep time (TST; bias: >90 min) and SEFF (bias: >13%) and underestimated wake after sleep onset (bias: >50 min). Sleep onset latency was accurately detected by the bedside radar (bias: 16 min). CSTs did not perform as well as actigraphy in estimating the all-night sleep summary measures. In an epoch-by-epoch concordance analysis, the bedside radar performed better in discriminating sleep versus wake (Matthew correlation coefficient [MCC]: mean 0.63, SD 0.12, 95% CI 0.57-0.69) than the undermattress devices (MCC of WSA: mean 0.41, SD 0.15, 95% CI 0.36-0.46; MCC of Emfit: mean 0.35, SD 0.16, 95% CI 0.26-0.43). The accuracy of identifying rapid eye movement and light sleep was poor across all CSTs, whereas deep sleep (ie, slow wave sleep) was predicted with moderate accuracy (MCC: >0.45) by both Somnofy and WSA. The deep sleep duration estimates of Somnofy correlated (r2=0.60; P
Alpha and theta oscillations characterize the waking human electroencephalogram (EEG) and can be modulated by closed-loop auditory stimulation (CLAS). These oscillations also occur during rapid eye movement (REM) sleep, but their function here remains elusive. CLAS represents a promising tool to pinpoint how these brain oscillations contribute to brain function in humans. Here we investigate whether CLAS can modulate alpha and theta oscillations during REM sleep in a phase-dependent manner. We recorded high-density EEG during an extended overnight sleep period in 18 healthy young adults. Auditory stimulation was delivered during both phasic and tonic REM sleep in alternating 6 s ON and 6 s OFF windows. During the ON windows, stimuli were phase-locked to four orthogonal phases of ongoing alpha or theta oscillations detected in a frontal electrode. The phases of ongoing alpha and theta oscillations were targeted with high accuracy during REM sleep. Alpha and theta CLAS induced phase-dependent changes in power and frequency at the target location. Frequency-specific effects were observed for alpha trough (speeding up) and rising (slowing down) and theta trough (speeding up) conditions. CLAS-induced phase-dependent changes were observed during both REM sleep substages, even though auditory evoked potentials were very much reduced in phasic compared to tonic REM sleep. This study provides evidence that faster REM sleep rhythms can be modulated by CLAS in a phase-dependent manner. This offers a new approach to investigate how modulation of REM sleep oscillations affects the contribution of this vigilance state to brain function.
Background: Sleep disorders are common among the aging population and people with neurodegenerative diseases. Sleep disorders have a strong bidirectional relationship with neurodegenerative diseases, where they accelerate and worsen one another. Although one-to-one individual cognitive behavioral interventions (conducted in-person or on the internet) have shown promise for significant improvements in sleep efficiency among adults, many may experience difficulties accessing interventions with sleep specialists, psychiatrists, or psychologists. Therefore, delivering sleep intervention through an automated chatbot platform may be an effective strategy to increase the accessibility and reach of sleep disorder intervention among the aging population and people with neurodegenerative diseases. Objective: This work aims to (1) determine the feasibility and usability of an automated chatbot (named MotivSleep) that conducts sleep interviews to encourage the aging population to report behaviors that may affect their sleep, followed by providing personalized recommendations for better sleep based on participants' self-reported behaviors; (2) assess the self-reported sleep assessment changes before, during, and after using our automated sleep disturbance intervention chatbot; (3) assess the changes in objective sleep assessment recorded by a sleep tracking device before, during, and after using the automated chatbot MotivSleep.Methods: We will recruit 30 older adult participants from West London for this pilot study. Each participant will have a sleep analyzer installed under their mattress. This contactless sleep monitoring device passively records movements, heart rate, and breathing rate while participants are in bed. In addition, each participant will use our proposed chatbot MotivSleep, accessible on WhatsApp, to describe their sleep and behaviors related to their sleep and receive personalized recommendations for better sleep tailored to their specific reasons for disrupted sleep. We will analyze questionnaire responses before and after the study to assess their perception of our proposed chatbot; questionnaire responses before, during, and after the study to assess their subjective sleep quality changes; and sleep parameters recorded by the sleep analyzer throughout the study to assess their objective sleep quality changes.Results: Recruitment will begin in May 2023 through UK Dementia Research Institute Care Research and Technology Centre organized community outreach. Data collection will run from May 2023 until December 2023. We hypothesize that participants will perceive our proposed chatbot as intelligent and trustworthy; we also hypothesize that our proposed chatbot can help improve participants' subjective and objective sleep assessment throughout the study.Conclusions: The MotivSleep automated chatbot has the potential to provide additional care to older adults who wish to improve their sleep in more accessible and less costly ways than conventional face-to-face therapy.
To compare the 24-hour sleep assessment capabilities of two contactless sleep technologies (CSTs) to actigraphy in community-dwelling older adults. We collected 7 to 14 days of data at home from 35 older adults (age: 65-83), some with medical conditions, using Withings Sleep Analyser (WSA, n=29), Emfit-QS (Emfit, n=17), a standard actigraphy device (Actiwatch Spectrum [AWS, n=34]) and a sleep diary. We compared nocturnal and daytime sleep measures estimated by the CSTs and actigraphy without sleep diary information (AWS-A) against sleep diary assisted actigraphy (AWS|SD). Compared to sleep diary, both CSTs accurately determined the timing of nocturnal sleep (ICC: going to bed, getting out of bed, time in bed > 0.75) whereas the accuracy of AWS-A was much lower. Compared to AWS|SD, the CSTs overestimated nocturnal total sleep time (WSA: +92.71±81.16 min; Emfit: +101.47±75.95 min) as did AWS-A (+46.95±67.26 min). The CSTs overestimated sleep efficiency (WSA: +9.19±14.26 %; Emfit: +9.41±11.05 %) whereas AWS-A estimate (-2.38±10.06 %) was accurate. About 65% (n=23) of participants reported daytime naps either in-bed or elsewhere. About 90% in-bed nap periods were accurately determined by WSA while Emfit was less accurate. All three devices estimated 24-h sleep duration with an error of ≈10% compared to the sleep diary. CSTs accurately capture the timing of in-bed nocturnal sleep periods without the need for sleep diary information. However, improvements are needed in assessing parameters such as total sleep time, sleep efficiency and naps before these CSTs can be fully utilized in field settings.
Sleep has been suggested to contribute to myelinogenesis and associated structural changes in the brain. As a principal hallmark of sleep, slow-wave activity (SWA) is homeostatically regulated but also differs between individuals. Besides its homeostatic function, SWA topography is suggested to reflect processes of brain maturation. Here, we assessed whether interindividual differences in sleep SWA and its homeostatic response to sleep manipulations are associated with in-vivo myelin estimates in a sample of healthy young men. Two hundred twenty-six participants (18-31 y.) underwent an in-lab protocol in which SWA was assessed at baseline (BAS), after sleep deprivation (high homeostatic sleep pressure, HSP) and sleep saturation (low homeostatic sleep pressure, LSP). Early-night frontal SWA, its frontal to occipital predominance as well as the overnight exponential SWA decay were computed over sleep conditions. Semi-quantitative magnetization transfer saturation maps (MTsat), providing markers for myelin content, were acquired during a separate laboratory visit. Early-night frontal SWA was negatively associated with regionally decreased myelin estimates in the temporal portion of the inferior longitudinal fasciculus. By contrast, neither the responsiveness of SWA to sleep saturation or deprivation, its overnight dynamics, nor the frontal/occipital SWA ratiowere associated with brain structural indices. Our results indicate that frontal SWA generation tracks interindividual differences in continued structural brain re-organization during early adulthood. This stage of life is not only characterized by ongoing region-specific changes in myelin content, but also by a sharp decrease and a shift towards frontal predominance in SWA generation.
Laboratory-based sleep manipulations show asymmetries between positive and negative affect, but say little about how more specific moods might change, and over what time course.. We report extensive analyses of items from the Positive and Negative Affect Scale (PANAS) during days following nights of chronic sleep restriction (6 hr sleep opportunity), during 40hr of acute sleep deprivation under constant routine conditions, and during a week-long forced desynchrony protocol in which participants lived on a 28-h day. Living in the laboratory resulted in medium effects sizes on all measures of positive affect (Attentiveness, General Positive Affect, Joviality, Assuredness), with a general deterioration as the day wore on. These effects were not found with negative moods. Sleep restriction reduced some positive affect, particularly Attentiveness (also General Positive), and increased Hostility. These effects were not found with negative affect. A burden of chronic sleep loss also led to lower positive affect when participants confronted the acute sleep loss challenge, and all positive affect, as well as Fearfulness, General Negative Affect and Hostility were affected. Sleeping at atypical circadian phases resulted mood change: all positive affect reduced, Hostility and General Negative Affect increased. Deteriorations increased the further participants slept from their typical nocturnal sleep. In most cases the changes induced by chronic or acute sleep loss or mis-time sleep waxed or waned across the waking day, with linear or various non-linear trends best fitting these time-awake-based changes. While extended laboratory stays do not emulate the fluctuating emotional demands of everyday living, these findings demonstrate that even in controlled settings mood changes systematically as sleep is shortened or mis-timed.
INTRODUCTION: Sleep disturbances are prevalent in Alzheimer’s disease (AD), but it is currently not known whether night-to-night variation in sleep predicts day-to-day variation in vigilance, cognition, mood, and behavior (daytime measures). METHODS: Subjective and objective sleep and daytime measures were collected daily for two weeks in 15 participants with mild AD, 8 mild cognitive impairment (MCI) and 22 with no cognitive impairment (NCI). Associations between daytime measures and four principal components of sleep (duration, quality, continuity and latency) were quantified using mixed-model regression. RESULTS: Sleepiness, alertness, contentedness, everyday memory errors, serial subtraction and behavioral problems were predicted by at least one of the components of sleep, and in particular sleep duration and continuity. Associations between variation in sleep and daytime measures were linear or quadratic and often different in AD from NCI. DISCUSSION: These findings imply that daytime functioning in AD may be improved by interventions that target sleep continuity.
Introduction Longitudinal monitoring of vital signs provides a method for identifying changes to general health in an individual and particularly so in older adults. The nocturnal sleep period provides a convenient opportunity to assess vital signs. Contactless technologies that can be embedded into the bedroom environment are unintrusive and burdenless and have the potential to enable seamless monitoring of vital signs. To realise this potential, these technologies need to be evaluated against gold standard measures and in relevant populations. Methods We evaluated the accuracy of heart rate and breathing rate measurements of three contactless technologies (two under-mattress trackers: Withings sleep analyser (WSA) and Emfit QS (Emfit) and a bedside radar: Somnofy) in a sleep laboratory environment and assessed their potential to capture vital signs (heart rate and breathing rate) in a real-world setting. Data were collected in 35 community dwelling older adults aged between 65 and 83 years (mean ± SD: 70.8 ± 4.9; 21 men) during a one-night clinical polysomnography (PSG) in a sleep laboratory, preceded by 7 to 14 days of data collection at-home. Several of the participants had health conditions including type-2 diabetes, hypertension, obesity, and arthritis and ≈49% (n = 17) had moderate to severe sleep apnea while ≈29% (n = 10) had periodic leg movement disorder. The under-mattress trackers provided estimates of both heart rate and breathing rate while the bedside radar provided only breathing rate. The accuracy of the heart rate and breathing rate estimated by the devices was compared to PSG electrocardiogram (ECG) derived heart rate (beats per minute, bpm) and respiratory inductance plethysmography thorax (RIP thorax) derived breathing rate (cycles per minute, cpm). We also evaluated breathing disturbance indices of snoring and the apnea-hypopnea index (AHI) available from the WSA. Results All three contactless technologies provided acceptable accuracy in estimating heart rate [mean absolute error (MAE) < 2.2 bpm and mean absolute percentage error (MAPE) < 5%] and breathing rate (MAE ≤ 1.6 cpm and MAPE < 12%) at 1 minute resolution. All three contactless technologies were able to capture changes in heart rate and breathing rate across the sleep period. The WSA snoring and breathing disturbance estimates were also accurate compared to PSG estimates (R-squared: WSA Snore: 0.76, p < 0.001; WSA AHI: 0.59, p < 0.001). Conclusion Contactless technologies offer an unintrusive alternative to conventional wearable technologies for reliable monitoring of heart rate, breathing rate, and sleep apnea in community dwelling older adults at scale. They enable assessment of night-to-night variation in these vital signs, which may allow the identification of acute changes in health, and longitudinal monitoring which may provide insight into health trajectories.
Evidence suggests that anthropogenic climate change is accelerating and is affecting human health globally. Despite urgent calls to address health effects in the context of the additional challenges of environmental degradation, biodiversity loss and ageing populations, the effects of climate change on specific health conditions are still poorly understood. Neurological diseases contribute substantially to the global burden of disease, and the possible direct and indirect consequences of climate change for people with these conditions are a cause for concern. Unaccustomed temperature extremes can impair the brain’s systems of resilience, thereby exacerbating or increasing susceptibility to neurological disease. In this Perspective, we explore how changing weather patterns resulting from climate change affect sleep — an essential restorative human brain activity, the quality of which is important for people with neurological diseases. We also consider the pervasive and complex influences of climate change on two common neurological conditions: stroke and epilepsy. We highlight the urgent need for research into the mechanisms underlying the effects of climate change on the brain in health and disease. We also discuss how neurologists can respond constructively to the climate crisis by raising awareness and promoting mitigation measures and research, actions which will bring widespread co-benefits.
Biological rhythms pervade physiology and pathophysiology across multiple timescales. Because of the limited sensing and algorithm capabilities of neuromodulation device technology to-date, insight into the influence of these rhythms on the efficacy of bioelectronic medicine has been infeasible. As the development of new devices begins to mitigate previous technology limitations, we propose that future devices should integrate chronobiological considerations in their control structures to maximize the benefits of neuromodulation therapy. We motivate this proposition with preliminary longitudinal data recorded from patients with Parkinson's disease and epilepsy during deep brain stimulation therapy, where periodic symptom biomarkers are synchronized to sub-daily, daily, and longer timescale rhythms. We suggest a physiological control structure for future bioelectronic devices that incorporates time-based adaptation of stimulation control, locked to patient-specific biological rhythms, as an adjunct to classical control methods and illustrate the concept with initial results from three of our recent case studies using chronotherapy-enabled prototypes. [Display omitted] Biological sciences; Neuroscience; Biotechnology; Bioelectronics
Sleep and circadian rhythm dysfunction is prevalent in schizophrenia, is associated with distress and poorer clinical status, yet remains an under-recognized therapeutic target. The development of new therapies requires the identification of the primary drivers of these abnormalities. Understanding of the regulation of sleep–wake timing is now sufficiently advanced for mathematical model-based analyses to identify the relative contribution of endogenous circadian processes, behavioral or environmental influences on sleep-wake disturbance and guide the development of personalized treatments. Here, we have elucidated factors underlying disturbed sleep-wake timing by applying a predictive mathematical model for the interaction of light and the circadian and homeostatic regulation of sleep to actigraphy, light, and melatonin profiles from 20 schizophrenia patients and 21 age-matched healthy unemployed controls, and designed interventions which restored sleep-circadian function. Compared to controls, those with schizophrenia slept longer, had more variable sleep timing, and received significantly fewer hours of bright light (light > 500 lux), which was associated with greater variance in sleep timing. Combining the model with the objective data revealed that non 24-h sleep could be best explained by reduced light exposure rather than differences in intrinsic circadian period. Modeling implied that late sleep offset and non 24-h sleep timing in schizophrenia can be normalized by changes in environmental light–dark profiles, without imposing major lifestyle changes. Aberrant timing and intensity of light exposure patterns are likely causal factors in sleep timing disturbances in schizophrenia. Implementing our new model-data framework in clinical practice could deliver personalized and acceptable light–dark interventions that normalize sleep-wake timing.
Several cellular pathways contribute to neurodegenerative tauopathy-related disorders. Microglial activation, a major component of neuroinflammation, is an early pathological hallmark that correlates with cognitive decline, while the unfolded protein response (UPR) contributes to synaptic pathology. Sleep disturbances are prevalent in tauopathies and may also contribute to disease progression. Few studies have investigated whether manipulations of sleep influence cellular pathological and behavioural features of tauopathy. We investigated whether trazodone, a licensed antidepressant with hypnotic efficacy in dementia, can reduce disease-related cellular pathways and improve memory and sleep in male rTg4510 mice with a tauopathy-like phenotype. In a 9-week dosing regimen, trazodone decreased microglial NLRP3 inflammasome expression and phosphorylated p38mitogen-activated protein kinase levels which correlated with the NLRP3 inflammasome, the UPR effector ATF4, and total tau levels. Trazodone reduced theta oscillations during REM sleep and enhanced rapid eye movement (REM) sleep duration. Olfactory memory transiently improved, and memory performance correlated with REM sleep duration and theta oscillations. These findings on the effects of trazodone on the NLRP3 inflammasome, the unfolded protein response and behavioural hallmarks of dementia warrant further studies on the therapeutic value of sleep-modulating compounds for tauopathies.
Analyses of gene-expression dynamics in research on circadian rhythms and sleep homeostasis often describe these two processes using separate models. Rhythmically expressed genes are, however, likely to be influenced by both processes. We implemented a driven, damped harmonic oscillator model to estimate the contribution of circadian- and sleep-wake-driven influences on gene expression. The model reliably captured a wide range of dynamics in cortex, liver, and blood transcriptomes taken from mice and humans under various experimental conditions. Sleep-wake-driven factors outweighed circadian factors in driving gene expression in the cortex, whereas the opposite was observed in the liver and blood. Because of tissueand gene-specific responses, sleep deprivation led to a long-lasting intra- and inter-tissue desynchronization. The model showed that recovery sleep contributed to these long-lasting changes. The results demonstrate that the analyses of the daily rhythms in gene expression must take the complex interactions between circadian and sleep-wake influences into account. A record of this paper's transparent peer review process is included in the supplemental information.
BACKGROUND: Twenty-four-hour rhythmicity in mammalian tissues and organs is driven by local circadian oscillators, systemic factors, the central circadian pacemaker, and light-dark cycles. At the physiological level, the neural and endocrine systems synchronize gene expression in peripheral tissues and organs to the twenty-four-hour day cycle, and disruption of such regulation has been shown to lead to pathological conditions. Thus, monitoring rhythmicity in tissues/organs holds promise for circadian medicine, however most tissues and organs are not easily accessible in humans and alternative approaches to quantify circadian rhythmicity are needed. We investigated the overlap between rhythmic transcripts in human blood and transcripts shown to be rhythmic in 64 tissues/organs of the baboon, how these rhythms are aligned with light-dark cycles and each other, and whether timing of tissue-specific rhythmicity can be predicted from a blood sample. RESULTS: We compared rhythmicity in transcriptomic time series collected from humans and baboons using set logic, circular cross-correlation, circular clustering, functional enrichment analyses and least squares regression. Of the 759 orthologous genes that were rhythmic in human blood, 652 (86%) were also rhythmic in at least one baboon tissue and most of these genes were associated with basic processes such as transcription and protein homeostasis. 109 (17%) of the 652 overlapping rhythmic genes were reported as rhythmic in only one baboon tissue or organ and several of these genes have tissue/organ-specific functions. The timing of human and baboon rhythmic transcripts displayed prominent ‘night’ and ‘day’ clusters, with genes in the dark cluster associated with translation. Alignment between baboon rhythmic transcriptomes and the overlapping human blood transcriptome was significantly closer when light onset, rather than midpoint of light, or end of light period, was used as phase reference point. The timing of overlapping human and baboon rhythmic transcriptomes was significantly correlated in 25 tissue/organs with an average earlier timing of 3.21 h (SD 2.47 h) in human blood. CONCLUSIONS: The human blood transcriptome contains sets of rhythmic genes that overlap with rhythmic genes of tissues/organs in baboon. The rhythmic sets vary across tissues/organs, but the timing of most rhythmic genes is similar in human blood and baboon tissues/organs. These results have implications for development of blood transcriptome-based biomarkers for circadian rhythmicity in tissues and organs.
Study Objectives. Sleep disturbances and genetic variants have been identified as risk factors for Alzheimer’s disease. Our goal was to assess whether genome-wide polygenic risk scores (PRS) for AD associate with sleep phenotypes in young adults, decades before typical AD symptom onset. Methods. We computed whole-genome Polygenic Risk Scores (PRS) for AD and extensively phenotyped sleep under different sleep conditions, including baseline sleep, recovery sleep following sleep deprivation and extended sleep opportunity, in a carefully selected homogenous sample of healthy 363 young men (22.1 y ± 2.7) devoid of sleep and cognitive disorders. Results. AD PRS was associated with more slow wave energy, i.e. the cumulated power in the 0.5-4 Hz EEG band, a marker of sleep need, during habitual sleep and following sleep loss, and potentially with large slow wave sleep rebound following sleep deprivation. Furthermore, higher AD PRS was correlated with higher habitual daytime sleepiness. Conclusions. These results imply that sleep features may be associated with AD liability in young adults, when current AD biomarkers are typically negative, and the notion that quantifying sleep alterations may be useful in assessing the risk for developing AD.
Cortisol is a robust circadian signal that synchronises peripheral circadian clocks with the central clock in the suprachiasmatic nucleus via glucocorticoid receptors that regulate peripheral gene expression. Misalignment of the cortisol rhythm with the sleep–wake cycle, as occurs in shift work, is associated with negative health outcomes, but underlying molecular mechanisms remain largely unknown. We experimentally induced misalignment between the sleep–wake cycle and melatonin and cortisol rhythms in humans and measured time series blood transcriptomics while participants slept in-phase and out-of-phase with the central clock. The cortisol rhythm remained unchanged, but many glucocorticoid signalling transcripts were disrupted by mistimed sleep. To investigate which factors drive this dissociation between cortisol and its signalling pathways, we conducted bioinformatic and temporal coherence analyses. We found that glucocorticoid signalling transcripts affected by mistimed sleep were enriched for binding sites for the transcription factor SP1. Furthermore, changes in the timing of the rhythms of SP1 transcripts, a major regulator of transcription, and changes in the timing of rhythms in transcripts of the glucocorticoid signalling pathways were closely associated. Associations between the rhythmic changes in factors that affect SP1 expression and its activity, such as STAT3, EP300, HSP90AA1, and MAPK1, were also observed. We conclude that plasma cortisol rhythms incompletely reflect the impact of mistimed sleep on glucocorticoid signalling pathways and that sleep–wake driven changes in SP1 may mediate disruption of these pathways. These results aid understanding of mechanisms by which mistimed sleep affects health.
Sleep disturbances and genetic variants have been identified as risk factors for Alzheimer's disease (AD). Our goal was to assess whether genome-wide polygenic risk scores (PRS) for AD associate with sleep phenotypes in young adults, decades before typical AD symptom onset. We computed whole-genome PRS for AD and extensively phenotyped sleep under different sleep conditions, including baseline sleep, recovery sleep following sleep deprivation, and extended sleep opportunity, in a carefully selected homogenous sample of 363 healthy young men (22.1 years ± 2.7) devoid of sleep and cognitive disorders. AD PRS was associated with more slow-wave energy, that is, the cumulated power in the 0.5-4 Hz EEG band, a marker of sleep need, during habitual sleep and following sleep loss, and potentially with larger slow-wave sleep rebound following sleep deprivation. Furthermore, higher AD PRS was correlated with higher habitual daytime sleepiness. These results imply that sleep features may be associated with AD liability in young adults, when current AD biomarkers are typically negative, and support the notion that quantifying sleep alterations may be useful in assessing the risk for developing AD.
Twenty-four-hour rhythms in physiology and behaviour are shaped by circadian clocks, environmental rhythms, and feedback of behavioural rhythms onto physiology. In space, 24 h signals such as those associated with the light-dark cycle and changes in posture, are weaker, potentially reducing the robustness of rhythms. Head down tilt (HDT) bed rest is commonly used to simulate effects of microgravity but how HDT affects rhythms in physiology has not been extensively investigated. Here we report effects of -6 degrees HDT during a 90-day protocol on 24 h rhythmicity in 20 men. During HDT, amplitude of light, motor activity, and wrist-temperature rhythms were reduced, evening melatonin was elevated, while cortisol was not affected during HDT, but was higher in the morning during recovery when compared to last session of HDT. During recovery from HDT, time in Slow-Wave Sleep increased. EEG activity in alpha and beta frequencies increased during NREM and REM sleep. These results highlight the profound effects of head-down-tilt-bed-rest on 24 h rhythmicity.
Accurate assessment of the intrinsic period of the human circadian pacemaker is essential for a quantitative understanding of how our circadian rhythms are synchronized to exposure to natural and man-made light-dark (LD) cycles. The gold standard method for assessing intrinsic period in humans is forced desynchrony (FD) which assumes that the confounding effect of lights-on assessment of intrinsic period is removed by scheduling sleep-wake and associated dim LD cycles to periods outside the range of entrainment of the circadian pacemaker. However, the observation that the mean period of free-running blind people is longer than the mean period of sighted people assessed by FD (24.50 0.17 h vs 24.15 0.20 h, 0.001) appears inconsistent with this assertion. Here, we present a mathematical analysis using a simple parametric model of the circadian pacemaker with a sinusoidal velocity response curve (VRC) describing the effect of light on the speed of the oscillator. The analysis shows that the shorter period in FD may be explained by exquisite sensitivity of the human circadian pacemaker to low light intensities and a VRC with a larger advance region than delay region. The main implication of this analysis, which generates new and testable predictions, is that current quantitative models for predicting how light exposure affects entrainment of the human circadian system may not accurately capture the effect of dim light. The mathematical analysis generates new predictions which can be tested in laboratory experiments. These findings have implications for managing healthy entrainment of human circadian clocks in societies with abundant access to light sources with powerful biological effects.
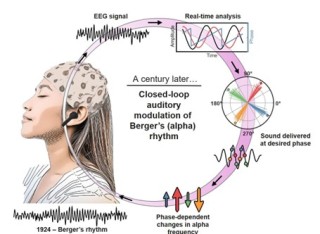
Alpha oscillations play a vital role in managing the brain's resources, inhibiting neural activity as a function of their phase and amplitude, and are changed in many brain disorders. Developing minimally invasive tools to modulate alpha activity and identifying the parameters that determine its response to exogenous modulators is essential for the implementation of focussed interventions. We introduce Alpha Closed-Loop Auditory Stimulation (αCLAS) as an EEG-based method to modulate and investigate these brain rhythms in humans with specificity and selectivity, using targeted auditory stimulation. Across a series of independent experiments, we demonstrate that αCLAS alters alpha power, frequency, and connectivity in a phase, amplitude, and topography-dependent manner. Using single-pulse-αCLAS, we show that the effects of auditory stimuli on alpha oscillations can be explained within the theoretical framework of oscillator theory and a phase-reset mechanism. Finally, we demonstrate the functional relevance of our approach by showing that αCLAS can interfere with sleep onset dynamics in a phase-dependent manner.
Data sources and code used for the analyses are found in the file called PLSR_16.zip which contains the following files and folders: File_contents_description.txt (detailed description of the files included in the zip file), TRAINING_SAMPLES_MicroarrayInformation.csv (description of the microarray samples conforming the training set), Training_Processed_SingleDataset.csv (pre-processed, normalised and filtered (i.e. ready to use) microarray data matrix conforming the training set), Training_TwoSamples_SamplingTable.csv (table describing the pairing of samples 12 hrs apart within the training set), VALIDATION_SAMPLES_MicroarrayInformation.csv (description of the microarray samples conforming the validation set), Validation_Processed_SingleDataset.csv (pre-processed, normalised and filtered (i.e. ready to use) microarray data matrix conforming the validation set), Validation_TwoSamples_SamplingTable.csv (table describing the pairing of samples 12 hrs apart within the validation set), Code (folder with R scripts (.R) described in Code/RUNNING_CODE_README.txt file). Further information about the methodology used can be found in the journal paper https://doi.org/10.7554/eLife.20214.
Training and validation processed datasets used in the analyses can be found in the file called SleepDebt.zip which contains the following folders: TimeAwake, SleepIncreaseDecrease, ChronicSleepInsufficiency and AcuteSleepLoss. Each folder contains R object files [.RData] with training and validation datasets, prediction labels and R2 in each set and lambda used in the regression analyses. Instrument- or software-specific information needed to interpret the data: R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. Further information about the methodology used can be found in the journal paper (Sleep, Volume 42, Issue 1, January 2019, zsy186).
Sleep disturbances have been reported as one of the most common symptoms among people living with dementia (PLWD). Few technologies to longitudinally monitor sleep across the 24-h day are available. Here, we develop machine learning models that use multimodal data to accurately monitor sleep across the day and night.
The daily alternation between sleep and wakefulness is one of the most dominant features of our lives and is a manifestation of the intrinsic 24 h rhythmicity underlying almost every aspect of our physiology. Circadian rhythms are generated by networks of molecular oscillators in the brain and peripheral tissues that interact with environmental and behavioural cycles to promote the occurrence of sleep during the environmental night. This alignment is often disturbed, however, by contemporary changes to our living environments, work or social schedules, patterns of light exposure, and biological factors, with consequences not only for sleep timing but also for our physical and mental health. Characterised by undesirable or irregular timing of sleep and wakefulness, in this Series paper we critically examine the existing categories of circadian rhythm sleep-wake disorders and the role of the circadian system in their development. We emphasise how not all disruption to daily rhythms is driven solely by an underlying circadian disturbance, and take a broader, dimensional approach to explore how circadian rhythms and sleep homoeostasis interact with behavioural and environmental factors. Very few high-quality epidemiological and intervention studies exist, and wider recognition and treatment of sleep timing disorders are currently hindered by a scarcity of accessible and objective tools for quantifying sleep and circadian physiology and environmental variables. We therefore assess emerging wearable technology, transcriptomics, and mathematical modelling approaches that promise to accelerate the integration of our knowledge in sleep and circadian science into improved human health.
Introduction Sleep plays a crucial role in brain plasticity, and has been suggested to be involved in myelin organization. Here we assessed the association between sleep homeostatic responses and quantitative MRI-derived myelin content in a sample of healthy young men. Methods: 238 male participants (age: 22.12.7) underwent an in-lab protocol to assess homeostatic responses in slow wave and REM sleep through a modulation of prior wakefulness and sleep duration. The protocol encompassed four conditions: a baseline night (BAS, duration adjusted on participant’s sleep-wake schedule), a 12h sleep extension night (EXT) followed by a 4-h nap and an 8-h sleep opportunity night (sleep saturation; SAT) and a 12h recovery night (REC) following 40-hours sleep deprivation. For each night, four sleep parameters were extracted: sleep slow wave activity at the beginning of the night (SWA0), its overnight exponential dissipation rate (tau), and overnight mean theta and beta power per REM epoch. Participants underwent a multiparameter brain MRI protocol at 3T to extract quantitative maps sensitive to different myelin biomarkers. F-contrasts were calculated to assess whether the modularity of sleep parameters across sleep conditions explains variance in myelin biomarkers. Reported statistics are family-wise-error corrected over the entire brain volume (pFWE BAS>EXT>SAT; all p < 0.001), while REM sleep percentage significantly differed only between SAT and the other sleep contexts (F(3,1257)= 13.676743, p
Sleep timing varies between individuals and can be altered in mental and physical health conditions. Sleep and circadian sleep phenotypes, including circadian rhythm sleep-wake disorders, may be driven by endogenous physiological processes, exogeneous environmental light exposure, social constraints or behavioural factors. Identifying the relative contributions of these driving factors to different phenotypes is essential for the design of personalised sleep interventions. The timing of the human sleep-wake cycle has been modelled as an interaction between a relaxation oscillator (the sleep homeostat), a stable limit cycle oscillator with a near 24-hour period (the circadian process), man-made light exposure, and the natural light-dark cycle generated by the Earth's rotation. However, these models have rarely been used to quantitatively describe sleep at the individual level. Here, we present a new Homeostatic-Circadian-Light model (HCL) which is simpler, more transparent, and more computationally efficient than other available models and is designed to run using longitudinal sleep and light exposure data from wearable sensors. We carry out a systematic sensitivity analysis for all model parameters and discuss parameter identifiability. We demonstrate we can describe individual sleep phenotypes in each of 34 older participants (65-83y) by feeding individual participant light exposure patterns into the model and fitting two parameters that capture individual average sleep duration and timing. The fitted parameters describe endogenous drivers of sleep phenotypes. We then quantify exogenous drivers using a novel metric which encodes the circadian phase dependence of the response to light. Combining endogenous and exogeneous drivers better explains individual mean mid-sleep (adjusted R-squared 0.64) than either driver on its own (adjusted R-squared 0.08 and 0.17 respectively). Critically, our model and analysis highlights different people exhibiting the same sleep phenotype may have different driving factors and opens the door to personalised interventions to regularize sleep-wake timing that are readily implementable with current digital health technology.Competing Interest StatementDJD and ACS are consultants to F. Hoffmann-La Roche, Ltd.Footnotes* This version of the manuscript has been revised to: - take account of referees comments - updated to correct minor errors, including typographical errors. The central messages and mathematical modelling remain unchanged.* https://github.com/anneskeldon/Homeostatic_circadian_light_model-factors_driving_sleep_phenotypes
Plasma biomarkers of dementia, including phosphorylated tau (p-tau217), offer promise as tools for diagnosis, stratification for clinical trials, monitoring disease progression, and assessing the success of interventions in those living with Alzheimer's disease. However, currently, it is unknown whether these dementia biomarker levels vary with the time of day, which could have implications for their clinical value. In two protocols, we studied 38 participants (70.8 ± 7.6 years; mean ± SD) in a 27-h laboratory protocol with either two samples taken 12 h apart or 3-hourly blood sampling for 24 h in the presence of a sleep-wake cycle. The study population comprised people living with mild Alzheimer's disease (PLWA, n = 8), partners/caregivers of PLWA (n = 6) and cognitively intact older adults (n = 24). Single-molecule array technology was used to measure phosphorylated tau (p-tau217) (ALZpath), amyloid-beta 40 (Aβ40), amyloid-beta 42 (Aβ42), glial fibrillary acidic protein, and neurofilament light (NfL) (Neuro 4-Plex E). Analysis with a linear mixed model (SAS, PROC MIXED) revealed a significant effect of time of day for p-tau217, Aβ40, Aβ42, and NfL, and a significant effect of participant group for p-tau217. For p-tau217, the lowest levels were observed in the morning upon waking and the highest values in the afternoon/early evening. The magnitude of the diurnal variation for p-tau217 was similar to the reported increase in p-tau217 over one year in amyloid-β-positive mild cognitively impaired people. Currently, the factors driving this diurnal variation are unknown and could be related to sleep, circadian mechanisms, activity, posture, or meals. Overall, this work implies that the time of day of sample collection may be relevant in the implementation and interpretation of plasma biomarkers in dementia research and care.
Abstract Background Increased daytime napping and excessive sleepiness are associated with cognitive decline in older adults, especially in people living with dementia (PLWD) [1]. Subjective assessments of naps are burdensome and maybe unreliable in PLWD and hence there is a need for technologies that provide objective longitudinal assessment of the incidence and duration of naps. Here we compare two contactless sleep technologies (CSTs) against sleep diary and actigraphy for monitoring daytime napping in community dwelling non‐demented older adults. Method Two under‐mattress CSTs (Withings Sleep Analyser [WSA] and Emfit QS [Emfit]) along with actigraphy (Actiwatch Spectrum [AWS]) were deployed in the home of 17 older adults for a period of 14 days ( = 65 years; Mean Age ± SD = 72 ± 4.49; 6 Women). The ground truth nap information was collected using an extended consensus sleep diary that included additional information about naps (timing, duration, and location). We analyzed the agreement of the napping events and duration estimated by WSA, Emfit and AWS against the sleep diary reported events. Result The CSTs only detected in‐bed naps whilst the AWS detected both in‐bed and not‐in‐bed naps. Although all the compared devices detected spurious naps unreported in the sleep diary, it was highest in AWS (81% of total naps detected) followed by Emfit (63%) and WSA (16%) as shown in Figure 1. Among the CSTs, WSA accurately detected more in bed naps while registering less spurious naps compared to Emfit but had lower duration agreement to sleep diary (Figure 2). Further, when the contribution of daytime naps to 24‐h total sleep time was computed, the WSA estimate (12.8±6.1%) was closest to the sleep diary estimate (12.9±9.1%) followed by AWS (15.6±9.9%) and Emfit (17.2±11.1%). Conclusion CSTs, with their ability to provide both contextual location information and objective measures of napping, such as timing and duration, offer a reliable and unobtrusive alternative to traditional methods such as sleep diary and actigraphy for long‐term round‐the‐clock monitoring of sleep in older adults. References: [1] Li, P, Gao, L, Yu, L, et al. Daytime napping, and Alzheimer’s dementia: A potential bidirectional relationship. Alzheimer’s Dement. 2023; 19: 158– 168. https://doi.org/10.1002/alz.12636
Period3 (Per3) is one of the most robustly rhythmic genes in humans and animals. It plays a significant role in temporal organisation in peripheral tissues. The effects of PER3 variants on many phenotypes have been investigated in targeted and genome-wide studies. PER3 variants, especially the human variable number tandem repeat (VNTR), associate with diurnal preference, mental disorders, non-visual responses to light, brain and cognitive responses to sleep loss/circadian misalignment. Introducing the VNTR into mice alters responses to sleep loss and expression of sleep homeostasis-related genes. Several studies were limited in size and some findings were not replicated. Nevertheless, the data indicate a significant contribution of PER3 to sleep and circadian phenotypes and diseases, which may be connected by common pathways. Thus, PER3-dependent altered light sensitivity could relate to high retinal PER3 expression and may contribute to altered brain response to light, diurnal preference and seasonal mood. Altered cognitive responses during sleep loss/circadian misalignment and changes to slow wave sleep may relate to changes in wake/activity-dependent patterns of hypothalamic gene expression involved in sleep homeostasis and neural network plasticity. Comprehensive characterisation of effects of clock gene variants may provide new insights into the role of circadian processes in health and disease.
Vigilance states, electroencephalogram (EEG) power spectra (0.25-25.0 Hz), and cortical temperature (TCRT) of 10 rats were obtained during a baseline day, a 24-h sleep deprivation (SD) period, and 2 days of recovery (recoveries 1 and 2). EEG power density in waking gradually increased in most frequencies during the SD period. Non-rapid-eye-movement (NREM) sleep was enhanced on both recovery days, and rapid-eye-movement sleep was enhanced only on recovery 1. In the initial 4 h of recovery 1, EEG slow-wave activity (SWA; mean power density 0.75-4.0 Hz) in NREM sleep was elevated relative to baseline, and the number of brief awakenings (nBA) was reduced. In the dark period of recovery 1 and the light period of recovery 2, SWA was below baseline, and nBA was increased. During the entire recovery period, SWA and nBA, both expressed as deviation from baseline values, were negatively correlated. During the SD period, TCRT was above baseline, and in the initial 16 h of recovery 1 it was below baseline. Whereas TCRT was negatively correlated with NREM sleep, no significant correlation was found between TCRT and SWA within NREM sleep. It is concluded that SD causes a short-lasting intensification of sleep, as indicated by the enhanced SWA and the reduced nBA, and a long-lasting increase in sleep duration. The different time courses of SWA and TCRT suggest that variations in NREM sleep intensity are not directly related to changes in TCRT.
In nine subjects sleep was recorded under base-line conditions with a habitual bedtime (prior wakefulness 16 h; lights off at 2300 h) and during recovery from sleep deprivation with a phase-advanced bedtime (prior wakefulness 36 h; lights off at 1900 h). The duration of phase-advanced recovery sleep was greater than 12 h in all subjects. Spectral analysis of the sleep electroencephalogram (EEG) revealed that slow-wave activity (SWA; 0.75-4.5 Hz) in non-rapid-eye-movement (NREM) sleep was significantly enhanced during the first two NREM-REM sleep cycles of displaced recovery sleep. The sleep stages 3 and 4 (slow-wave sleep) and SWA decreased monotonically over the first three and four NREM-REM cycles of, respectively, base-line and recovery sleep. The time course of SWA in base-line and recovery sleep could be adequately described by an exponentially declining function with a horizontal asymptote. The results are in accordance with the two-process model of sleep regulation in which it is assumed that SWA rises as a function of the duration of prior wakefulness and decreases exponentially as a function of prior sleep. We conclude that the present data do not provide evidence for a 12.5-h sleep-dependent rhythm of deep NREM sleep.
Objective: Sleep monitoring has extensively utilized electroencephalogram (EEG) data collected from the scalp, yielding very large data repositories and well-trained analysis models. Yet, this wealth of data is lacking for emerging, less intrusive modalities, such as ear-EEG.Methods and procedures: The current study seeks to harness the abundance of open-source scalp EEG datasets by applying models pre-trained on data, either directly or with minimal fine-tuning; this is achieved in the context of effective sleep analysis from ear-EEG data that was recorded using a single in-ear electrode, referenced to the ipsilateral mastoid, and developed in-house as described in our previous work. Unlike previous studies, our research uniquely focuses on an older cohort (17 subjects aged 65-83, mean age 71.8 years, some with health conditions), and employs LightGBM for transfer learning, diverging from previous deep learning approaches. Results: Results show that the initial accuracy of the pre-trained model on ear-EEG was 70.1%, but fine-tuning the model with ear-EEG data improved its classification accuracy to 73.7%. The fine-tuned model exhibited a statistically significant improvement (p < 0.05, dependent t-test) for 10 out of the 13 participants, as reflected by an enhanced average Cohen's kappa score (a statistical measure of inter-rater agreement for categorical items) of 0.639, indicating a stronger agreement between automated and expert classifications of sleep stages. Comparative SHAP value analysis revealed a shift in feature importance for the N3 sleep stage, underscoring the effectiveness of the fine-tuning process.Conclusion: Our findings underscore the potential of fine-tuning pre-trained scalp EEG models on ear-EEG data to enhance classification accuracy, particularly within an older population and using feature-based methods for transfer learning. This approach presents a promising avenue for ear-EEG analysis in sleep studies, offering new insights into the applicability of transfer learning across different populations and computational techniques.Clinical impact: An enhanced ear-EEG method could be pivotal in remote monitoring settings, allowing for continuous, non-invasive sleep quality assessment in elderly patients with conditions like dementia or sleep apnea.
This British Association for Psychopharmacology guideline replaces the original version published in 2010, and contains updated information and recommendations. A consensus meeting was held in London in October 2017 attended by recognised experts and advocates in the field. They were asked to provide a review of the literature and identification of the standard of evidence in their area, with an emphasis on meta-analyses, systematic reviews and randomised controlled trials where available, plus updates on current clinical practice. Each presentation was followed by discussion, aiming to reach consensus where the evidence and/or clinical experience was considered adequate, or otherwise to flag the area as a direction for future research. A draft of the proceedings was circulated to all speakers for comments, which were incorporated into the final statement.
•Sleep is a key concern in dementias but their sleep phenotypes are not well defined.•We addressed this issue in major FTD and AD syndromes versus healthy older controls.•We surveyed sleep duration, quality and disruptive events, and daytime somnolence.•Sleep symptoms were frequent in FTD and AD and distinguished these diseases.•Sleep disturbance is an important clinical issue across major FTD and AD syndromes. Sleep disruption is a key clinical issue in the dementias but the sleep phenotypes of these diseases remain poorly characterised. Here we addressed this issue in a proof-of-principle study of 67 patients representing major syndromes of frontotemporal dementia (FTD) and Alzheimer’s disease (AD), in relation to 25 healthy older individuals. We collected reports on clinically-relevant sleep characteristics - time spent overnight in bed, sleep quality, excessive daytime somnolence and disruptive sleep events. Difficulty falling or staying asleep at night and excessive daytime somnolence were significantly more frequently reported for patients with both FTD and AD than healthy controls. On average, patients with FTD and AD retired earlier and patients with AD spent significantly longer in bed overnight than did healthy controls. Excessive daytime somnolence was significantly more frequent in the FTD group than the AD group; AD syndromic subgroups showed similar sleep symptom profiles while FTD subgroups showed more variable profiles. Sleep disturbance is a significant clinical issue in major FTD and AD variant syndromes and may be even more salient in FTD than AD. These preliminary findings warrant further systematic investigation with electrophysiological and neuroanatomical correlation in major proteinopathies.
Disturbances of the sleep-wake cycle are highly prevalent and diverse. The aetiology of some sleep disorders, such as circadian rhythm sleep-wake disorders, is understood at the conceptual level of the circadian and homeostatic regulation of sleep and in part at a mechanistic level. Other disorders such as insomnia are more difficult to relate to sleep regulatory mechanisms or sleep physiology. To further our understanding of sleep-wake disorders and the potential of novel therapeutics, we discuss recent findings on the neurobiology of sleep regulation and circadian rhythmicity and its relation with the subjective experience of sleep and the quality of wakefulness. Sleep continuity and to some extent REM sleep emerge as determinants of subjective sleep quality and waking performance. The effects of insufficient sleep primarily concern subjective and objective sleepiness as well as vigilant attention, whereas performance on higher cognitive functions appears to be better preserved albeit at the cost of increased effort. We discuss age-related, sex and other trait-like differences in sleep physiology and sleep need and compare the effects of existing pharmacological and non-pharmacological sleep- and wake-promoting treatments. Successful non-pharmacological approaches such as sleep restriction for insomnia and light and melatonin treatment for circadian rhythm sleep disorders target processes such as sleep homeostasis or circadian rhythmicity. Most pharmacological treatments of sleep disorders target specific signalling pathways with no well-established role in either sleep homeostasis or circadian rhythmicity. Pharmacological sleep therapeutics induce changes in sleep structure and the sleep EEG which are specific to the mechanism of action of the drug. Sleep- and wake-promoting therapeutics often induce residual effects on waking performance and sleep, respectively. The need for novel therapeutic approaches continues not at least because of the societal demand to sleep and be awake out of synchrony with the natural light-dark cycle, the high prevalence of sleep-wake disturbances in mental health disorders and in neurodegeneration. Novel approaches, which will provide a more comprehensive description of sleep and allow for large-scale sleep and circadian physiology studies in the home environment, hold promise for continued improvement of therapeutics for disturbances of sleep, circadian rhythms and waking performance.
Sleep in mammals consists of non-rapid-eye-movement and rapid-eye movement sleep. A large genetic screen reveals that these two sleep states are altered in mice by mutations dubbed Sleepy and Dreamless. SEE ARTICLE p.378
SUMMARY Night work is associated with increased sleepiness and disturbed sleep. Maladaptation of the circadian system, which is phase‐adjusted to day time work and thus promotes sleepiness during its nadir at night and wakefulness (or disturbed sleep) during the day, contributes substantially to this problem. A major cause of suboptimal circadian phase adjustment among night workers is the exposure to morning light, which prevents the delay needed for optimal adjustment to night work. Several laboratory studies indicate that careful application of bright light may cause the circadian system to shift to any desired phase. Furthermore, studies of simulated night work demonstrate that night exposure to bright light can virtually eliminate circadian maladjustment among night workers. While the results are promising, there is still, however, an urgent need for longitudinal studies of bright light application in. real‐life settings.
Abstract Adrenal glucocorticoids are major modulators of multiple functions, including energy metabolism, stress responses, immunity, and cognition. The endogenous secretion of glucocorticoids is normally characterized by a prominent and robust circadian (around 24 hours) oscillation, with a daily peak around the time of the habitual sleep-wake transition and minimal levels in the evening and early part of the night. It has long been recognized that this 24-hour rhythm partly reflects the activity of a master circadian pacemaker located in the suprachiasmatic nucleus of the hypothalamus. In the past decade, secondary circadian clocks based on the same molecular machinery as the central master pacemaker were found in other brain areas as well as in most peripheral tissues, including the adrenal glands. Evidence is rapidly accumulating to indicate that misalignment between central and peripheral clocks has a host of adverse effects. The robust rhythm in circulating glucocorticoid levels has been recognized as a major internal synchronizer of the circadian system. The present review examines the scientific foundation of these novel advances and their implications for health and disease prevention and treatment.
Advances in diagnostic technology, including chronic intracranial EEG recordings, have confirmed the clinical observation of different temporal patterns of epileptic activity and seizure occurrence over a 24-h period. The rhythmic patterns in epileptic activity and seizure occurrence are probably related to vigilance states and circadian variation in excitatory and inhibitory balance. Core circadian genes BMAL1 and CLOCK, which code for transcription factors, have been shown to influence excitability and seizure threshold. Despite uncertainties about the relative contribution of vigilance states versus circadian rhythmicity, including circadian factors such as seizure timing improves sensitivity of seizure prediction algorithms in individual patients. Improved prediction of seizure occurrence opens the possibility for personalised antiepileptic drug-dosing regimens timed to particular phases of the circadian cycle to improve seizure control and to reduce side-effects and risks associated with seizures. Further studies are needed to clarify the pathways through which rhythmic patterns of epileptic activity are generated, because this might also inform future treatment options.
Sleep timing and sleep structure vary between and within individuals and are regulated by two main processes: sleep homeostasis and circadian rhythmicity. Homeostasis refers to the ability of an organism to maintain an internal biologic equilibrium through regulatory mechanisms. Homeostatic regulation of sleep has been demonstrated by driving the system away from a state of equilibrium through total, partial, and sleep stage specific deprivation and then monitoring the resultant changes in sleep. The homeostatic regulation of slow-wave activity has been quantified in detail but other aspects of sleep, such as sleep duration and REM sleep, are also under homeostatic control. All these aspects of sleep are also affected by circadian rhythmicity. The joint regulation of sleep timing by homeostasis and circadian rhythmicity has been formalised in the two-process model of sleep regulation. More recently, effects of light on the human circadian pacemaker have been incorporated in models of sleep regulation and other models developed to describe the ultradian NREM-REM cycle. Conceptual models of sleep regulation summarise accumulated knowledge and extract essential principles underlying empirical facts. Mathematical models can in addition test whether our understanding of phenomena as formulated in conceptual frameworks is sufficient to explain these phenomena quantitatively. Ultimately, models for sleep regulation should help us to understand sleep phenotypes, treat sleep disturbances, design physical and social environments to maximise the beneficial effects of sleep, and inform the development of policies to minimise the negative effects of insufficient and mistimed sleep.
Sleep is a process of rest and renewal that is vital for humans. However, there are several sleep disorders such as rapid eye movement (REM) sleep behaviour disorder (RBD), sleep apnea, and restless leg syndrome (RLS) that can have an impact on a significant portion of the population. These disorders are known to be associated with particular behaviours such as specific body positions and movements. Clinical diagnosis requires patients to undergo polysomnography (PSG) in a sleep unit as a gold standard assessment. This involves attaching multiple electrodes to the head and body. In this experiment, we seek to develop non-contact approach to measure sleep disorders related to body postures and movement. An Infrared (IR) camera is used to monitor body position unaided by other sensors. Twelve participants were asked to adopt and then move through a set of 12 pre-defined sleep positions. We then adopted convolutional neural networks (CNNs) for automatic feature generation from IR data for classifying different sleep postures. The results show that the proposed method has an accuracy of between 0.76 & 0.91 across the participants and 12 sleep poses with, and without a blanket cover, respectively. The results suggest that this approach is a promising method to detect common sleep postures and potentially characterise sleep disorder behaviours.
Introduction Disturbances of sleep/wake behaviour are amongst the most disabling symptoms of dementia, leading to increased carers’ burden and institutionalisation. The lack of unobtrusive, low- burden technologies validated to monitor sleep in patients living with dementia (PLWD) has prevented longitudinal studies of nocturnal disturbances and their correlates. Aims To examine the effect of medication changes and clinical status on the intraindividual variation in sleep/wake behaviour in PLWD. Methods Using under-mattress pressure-sensing mat in 46 PLWD, we monitored sleep/wake behavioural metrics for 13,711 nights between 2019-2021. Machine learning and >3.6million nightly summaries from 13,671 individuals from the general population were used to detect abnormalities in PLWD’s nightly sleep/wake metrics and convert them to risk scores. Additionally, GP records were reviewed for each patient to determine whether medication changes and clinical events affected sleep parameters. Results PLWD’s went to bed earlier and rose later than sex- and age-matched controls. They had more nocturnal awakenings with longer out-of-bed durations. Notably, at the individual patient level, increased metric-specific risk scores were temporally related to changes in antipsychotics and antidepressants, and acute illness, including UTI, cardiac events, and depressive episodes. Conclusions Passive monitoring of sleep/wake behaviours is a promising way to identify novel markers of disease progression and evaluate the effectiveness of pharmaceutical interventions in patients with dementia.
Timing of the human sleep-wake cycle is determined by social constraints, biological processes (sleep homeostasis and circadian rhythmicity) and environmental factors, particularly natural and electrical light exposure. To what extent seasonal changes in the light-dark cycle affect sleep timing and how this varies between weekdays and weekends has not been firmly established. We examined sleep and activity patterns during weekdays and weekends in late autumn (standard time, ST) and late spring (daylight saving time, DST), and expressed their timing in relation to three environmental reference points: clock-time, solar noon (SN), which occurs one clock hour later during DST than ST, and the midpoint of accumulated light exposure (50%-LE). Observed sleep timing data were compared to simulated data from a mathematical model for the effects of light on the circadian and homeostatic regulation of sleep. A total of 715 days of sleep timing and light exposure were recorded in 19 undergraduates in a repeated-measures observational study. During each three-week assessment, light and activity were monitored, and self-reported bed and wake times were collected. Light exposure was higher in spring than in autumn. 50%-LE did not vary across season, but occurred later on weekends compared to weekdays. Relative to clock-time, bedtime, wake-time, mid-sleep, and midpoint of activity were later on weekends but did not differ across seasons. Relative to SN, sleep and activity measures were earlier in spring than in autumn. Relative to 50%-LE, only wake-time and mid-sleep were later on weekends, with no seasonal differences. Individual differences in mid-sleep did not correlate with SN but correlated with 50%-LE. Individuals with different habitual bedtimes responded similarly to seasonal changes. Model simulations showed that light exposure patterns are sufficient to explain sleep timing in spring but less so in autumn. The findings indicate that during autumn and spring, the timing of sleep associates with actual light exposure rather than sun time as indexed by SN.
A common complaint of older persons is disturbed sleep, typically characterized as an inability to return to sleep after waking. As every sleep episode (i.e., time in bed) includes multiple transitions between wakefulness and sleep (which can be subdivided into rapid eye movement [REM] sleep and non-REM [NREM] sleep), we applied survival analysis to sleep data to determine whether changes in the "hazard" (duration-dependent probability) of awakening from sleep and/or returning to sleep underlie age-related sleep disturbances. The hazard of awakening from sleep-specifically NREM sleep-was much greater in older than in young adults. We found, however, that when an individual had spontaneously awakened, the probability of falling back asleep was not greater in young persons. Independent of bout length, the number of transitions between NREM and REM sleep stages relative to number of transitions to wake was approximately 6 times higher in young than older persons, highlighting the difficulty in maintaining sleep in older persons. Interventions to improve age-related sleep complaints should thus target this change in awakenings. © 2013 Elsevier Inc.
Local and national governments around the world are currently considering the elimination of the annual switch to and from Daylight Saving Time (DST). As an international organization of scientists dedicated to studying circadian and other biological rhythms, the Society for Research on Biological Rhythms (SRBR) engaged experts in the field to write a Position Paper on the consequences of choosing to live on DST or Standard Time (ST). The authors take the position that, based on comparisons of large populations living in DST or ST or on western versus eastern edges of time zones, the advantages of permanent ST outweigh switching to DST annually or permanently. Four peer reviewers provided expert critiques of the initial submission, and the SRBR Executive Board approved the revised manuscript as a Position Paper to help educate the public in their evaluation of current legislative actions to end DST.
The brain has a unique macroscopic waste clearance system, termed the glymphatic system which utilises perivascular tunnels surrounded by astroglia to promote cerebrospinal-interstitial fluid exchange. Rodent studies have demonstrated a marked increase in glymphatic clearance during sleep which has been linked to a sleep-induced expansion of the extracellular space and concomitant reduction in intracellular volume. However, despite being implicated in the pathophysiology of multiple human neurodegenerative disorders, non-invasive techniques for imaging glymphatic clearance in humans are currently limited. Here we acquired multi-shell diffusion weighted MRI (dwMRI) in twenty-one healthy young participants (6 female, 22.3 ± 3.2 years) each scanned twice, once during wakefulness and once during sleep induced by a combination of one night of sleep deprivation and 10mg of the hypnotic zolpidem 30 minutes before scanning. To capture hypothesised sleep-associated changes in intra/extracellular space, dwMRI were analysed using higher order diffusion modelling with the prediction that sleep-associated increases in interstitial (extracellular) fluid volume would result in a decrease in diffusion kurtosis, particularly in areas associated with slow wave generation at the onset of sleep. In line with our hypothesis, we observed a global reduction in diffusion kurtosis (t15=2.82, p=0.006) during sleep as well as regional reductions in brain areas associated with slow wave generation during early sleep and default mode network areas that are highly metabolically active during wakefulness. Analysis with a higher-order representation of diffusion (MAP-MRI) further indicated that changes within the intra/extracellular domain rather than membrane permeability likely underpin the observed sleep-associated decrease in kurtosis. These findings identify higher-order modelling of dwMRI as a potential new non-invasive method for imaging glymphatic clearance and extend rodent findings to suggest that sleep is also associated with an increase in interstitial fluid volume in humans.
Sleep is essential for the maintenance of human life, yet many features of sleep are poorly understood and mathematical models are an important tool for probing proposed biological mechanisms. The most well-known mathematical model of sleep regulation, the two-process model, models the sleep-wake cycle by two oscillators: a circadian oscillator and a homeostatic oscillator. An alternative, more recent, model considers the reciprocal interaction of sleep promoting neurons and the ascending arousal system regulated by homeostatic and circadian processes. Here we show there are fundamental similarities between these two models. The implications are illustrated with two important sleep-wake phenomena. Firstly, we show that in the two-process model, transitions between different numbers of daily sleep episodes can be classified as grazing bifurcations. This provides the theoretical underpinning for numerical results showing that the sleep patterns of many mammals can be explained by the reciprocal interaction model. Secondly, we show that when sleep deprivation disrupts the sleep-wake cycle, ostensibly different measures of sleepiness in the two models are closely related. The demonstration of the mathematical similarities of the two models is important because not only does it it allow some features of the two-process model to be interpreted physiologically but it also means that knowledge gained from the study of the two-process model can be used to inform understanding of the behaviour of the mutual inhibition model. This is important because the mutual inhibition model and its extensions are increasingly being used as a tool to understand a diverse range of sleep-wake phenomena sucah as the design of optimal shift-patterns, yet the values it uses for the parameters associated with the circadian and homeostatic processes are very different from those that have been experimentally measured in the context of the two-process model
Background Sleep disturbance is common among people living with dementia as well as their caregivers. Non‐contact video technology can be used to characterise such disturbances as well as quantifying sleep quality by measuring the number of sleep body positions (poses). Such an approach may be beneficial for home‐based longitudinal clinical monitoring of sleep pattern changes and disturbances at all stages of dementia. Here we present our pilot results of a personalised data‐driven method applied to video data for quantification of sleep disturbance comparing older and younger participants. Method Data were collected in two separate studies which included an overnight 10‐12 hour laboratory sleep recording from thirteen older (65‐80 years, 9 male:4 female) and eleven younger (18‐34 years, 7 male:4 female) participants in a dedicated sleep facility. A data‐driven analysis using Principal Component Analysis and k‐means clustering was applied to infrared video data extracted from a clinical polysomnography (PSG) system. The data‐driven analysis automatically determined statistically significant groupings or clusters of unique body poses for each individual. Pose number, number of pose transitions, pose duration, and pose transition duration were computed for each participant. Result The number of data‐driven poses in older and younger participants was remarkably similar with 15.2±3.9 and 15.8±1.8 (mean±SD) poses per participant, respectively. However, the older group had a higher number of pose transitions (33.0±8.82) compared to the younger group (23.9±6.83) (p = 0.03). A significant 20% difference (p = 0.03) in the average duration of each body position was observed, with 62.3±7.1 minutes and 78.9±15.9 minutes for the older and younger groups, respectively (see Figure 1 and Figure 2). Pose transition duration was 19.2±7.25 seconds and 14.6±4.32 seconds for the older and younger groups, respectively where they were not significantly different. Conclusion Although the number of body positions did not vary significantly between the two cohorts, the older group changed body position more frequently and it took them longer to do so. Data‐driven automated analysis of video‐based sleep monitoring holds significant promise for quantifying age‐related and inter‐individual differences in sleep behavior.
Nocturnal disturbance is frequently observed in dementia and is a major contributor to institutionalisation. Unobtrusive technology that can quantify sleep/wake and determine bed occupancy during the major nocturnal sleep episode may be beneficial for long-term clinical monitoring and the carer. Such technologies have, however, not been validated in older people. Here we assessed the performance of the Withings Sleep Mattress (WSM) in a heterogenous older population to ensure external validity.
Wearable heart rate monitors offer a cost-effective way of non-invasive, long-term monitoring of cardiac health. Validation of wearable technologies in an older populations is essential for evaluating their effectiveness during deployment in healthcare settings. To this end, we evaluated the validity of heart rate measures from a wearable device, Empatica E4, and compared them to the electrocardiography (ECG). We collected E4 data simultaneously with ECG in thirty-five older men and women during an overnight sleep recording in the laboratory. We propose a robust approach to resolve the missing inter-beat interval (IBI) data and improve the quality of E4 derived measures. We also evaluated the concordance of heart rate (HR) and heart rate variability (HRV) measures with ECG. The results demonstrate that the automatic E4 heart rate measures capture long-term HRV whilst the short-term metrics are affected by missing IBIs. Our approach provides an effective way to resolve the missing IBI issue of E4 and extracts reliable heart rate measures that are concordant with ECG. Clinical Relevance— This work discusses data quality challenges in heart rate data acquired by wearables and provides an efficient and reliable approach for extracting heart rate measures from the E4 wearable device and validates the metrics in older adults
Sleep complaints and irregular sleep patterns, such as curtailed sleep during workdays and longer and later sleep during weekends, are common. It is often implied that differences in circadian period and in entrained phase contribute to these patterns, but few data are available. We assessed parameters of the circadian rhythm of melatonin at baseline and in a forced desynchrony protocol in 35 participants (18 women) with no sleep disorders. Circadian period varied between 23 h 50 min and 24 h 31 min, and correlated positively (n = 31, rs = 0.43, P = 0.017) with the timing of the melatonin rhythm relative to habitual bedtime. The phase of the melatonin rhythm correlated with the Insomnia Severity Index (n = 35, rs = 0.47, P = 0.004). Self-reported time in bed during free days also correlated with the timing of the melatonin rhythm (n = 35, rs = 0.43, P = 0.01) as well as with the circadian period (n = 31, rs = 0.47, P = 0.007), such that individuals with a more delayed melatonin rhythm or a longer circadian period reported longer sleep during the weekend. The increase in time in bed during the free days correlated positively with circadian period (n = 31, rs = 0.54, P = 0.002). Polysomnographically assessed latency to persistent sleep (n = 34, rs = 0.48, P = 0.004) correlated with the timing of the melatonin rhythm when participants were sleeping at their habitual bedtimes in the laboratory. This correlation was significantly stronger in women than in men (Z = 2.38, P = 0.017). The findings show that individual differences in circadian period and phase of the melatonin rhythm associate with differences in sleep, and suggest that individuals with a long circadian period may be at risk of developing sleep problems.
Quantification of sleep is important for the diagnosis of sleep disorders and sleep research. However, the only widely accepted method to obtain sleep staging is by visual analysis of polysomnography (PSG), which is expensive and time consuming. Here, we investigate automated sleep scoring based on a low‐cost, mobile electroencephalogram (EEG) platform consisting of a lightweight EEG amplifier combined with flex‐printed cEEGrid electrodes placed around the ear, which can be implemented as a fully self‐applicable sleep system. However, cEEGrid signals have different amplitude characteristics to normal scalp PSG signals, which might be challenging for visual scoring. Therefore, this study evaluates the potential of automatic scoring of cEEGrid signals using a machine learning classifier (“random forests”) and compares its performance with manual scoring of standard PSG. In addition, the automatic scoring of cEEGrid signals is compared with manual annotation of the cEEGrid recording and with simultaneous actigraphy. Acceptable recordings were obtained in 15 healthy volunteers (aged 35 ± 14.3 years) during an extended nocturnal sleep opportunity, which induced disrupted sleep with a large inter‐individual variation in sleep parameters. The results demonstrate that machine‐learning‐based scoring of around‐the‐ear EEG outperforms actigraphy with respect to sleep onset and total sleep time assessments. The automated scoring outperforms human scoring of cEEGrid by standard criteria. The accuracy of machine‐learning‐based automated scoring of cEEGrid sleep recordings compared with manual scoring of standard PSG was satisfactory. The findings show that cEEGrid recordings combined with machine‐learning‐based scoring holds promise for large‐scale sleep studies.
The transition from sleep to wakefulness entails a temporary period of reduced alertness and impaired performance known as sleep inertia. The extent to which its severity varies with task and cognitive processes remains unclear. We examined sleep inertia in alertness, attention, working memory and cognitive throughput with the Karolinska Sleepiness Scale (KSS), the Psychomotor Vigilance Task (PVT), n-back and add tasks, respectively. The tasks were administered 2 hours before bedtime and at regular intervals for four hours, starting immediately after awakening in the morning, in eleven participants, in a four-way cross-over laboratory design. We also investigated whether exposure to Blue-Enhanced or Bright Blue-Enhanced white light would reduce sleep inertia. Alertness and all cognitive processes were impaired immediately upon awakening (p
The power of the application of bioinformatics across multiple publicly available transcriptomic data sets was explored. Using 19 human and mouse circadian transcriptomic data sets, we found that NR1D1 and NR1D2 which encode heme-responsive nuclear receptors are the most rhythmic transcripts across sleep conditions and tissues suggesting that they are at the core of circadian rhythm generation. Analyzes of human transcriptomic data show that a core set of transcripts related to processes including immune function, glucocorticoid signalling, and lipid metabolism is rhythmically expressed independently of the sleep-wake cycle. We also identify key transcripts associated with transcription and translation that are disrupted by sleep manipulations, and through network analysis identify putative mechanisms underlying the adverse health outcomes associated with sleep disruption, such as diabetes and cancer. Comparative bioinformatics applied to existing and future data sets will be a powerful tool for the identification of core circadian- and sleep-dependent molecules.
Human performance results from an interaction between circadian rhythmicity and homeostatic sleep pressure. Whether and how this interaction is represented at the regional brain level is not established. We quantified changes in brain responses to a sustained-attention task during 13 functional magnetic resonance imaging (fMRI) sessions scheduled across the circadian cycle during 42h of wakefulness and following recovery sleep, in 33 healthy participants. Cortical responses showed significant circadian rhythmicity, the phase of which varied across brain regions. Cortical responses also significantly decreased with accrued sleep debt. Subcortical areas exhibited primarily a circadian modulation, which closely followed the melatonin profile. These findings expand our understanding of the mechanisms involved in maintaining cognition during the day and its deterioration during sleep deprivation and circadian misalignment.
Sleep and its sub-states are assumed to be important for brain function across the lifespan but which aspects of sleep associate with various aspects of cognition, mood and self-reported sleep quality has not yet been established in detail. Sleep was quantified by polysomnography, quantitative Electroencephalogram (EEG) analysis and self-report in 206 healthy men and women, aged 20–84 years, without sleep complaints. Waking brain function was quantified by five assessments scheduled across the day covering objectively assessed performance across cognitive domains including sustained attention and arousal, decision and response time, motor and sequence control, working memory, and executive function as well as self-reports of alertness, mood and affect. Controlled for age and sex, self-reported sleep quality was negatively associated with number of awakenings and positively associated with the duration of Rapid Eye Movement (REM) sleep, but no significant associations with Slow Wave Sleep (SWS) measures were observed. Controlling only for age showed that associations between objective and subjective sleep quality were much stronger in women than in men. Analysis of 51 performance measures demonstrated that, after controlling for age and sex, fewer awakenings and more REM sleep were associated significantly with better performance on the Goal Neglect task, which is a test of executive function. Factor analysis of the individual performance measures identified four latent variables labeled Mood/Arousal, Response Time, Accuracy, and Visual Perceptual Sensitivity. Whereas Mood/Arousal improved with age, Response Times became slower, while Accuracy and Visual perceptual sensitivity showed little change with age. After controlling for sex and age, nominally significant association between sleep and factor scores were observed such that Response Times were faster with more SWS, and Accuracy was reduced where individuals woke more often or had less REM sleep. These data identify a positive contribution of SWS to processing speed and in particular highlight the importance of sleep continuity and REM sleep for subjective sleep quality and performance accuracy across the adult lifespan. These findings warrant further investigation of the contribution of sleep continuity and REM sleep to brain function.
Sleep and circadian rhythm disturbance are predictors of poor physical and mental health, including dementia. Long-term digital technology-enabled monitoring of sleep and circadian rhythms in the community has great potential for early diagnosis, monitoring of disease progression, and assessing the effectiveness of interventions. Before novel digital technology-based monitoring can be implemented at scale, its performance and acceptability need to be evaluated and compared to gold-standard methodology in relevant populations. Here, we describe our protocol for the evaluation of novel sleep and circadian technology which we have applied in cognitively intact older adults and are currently using in people living with dementia (PLWD). In this protocol, we test a range of technologies simultaneously at home (7-14 days) and subsequently in a clinical research facility in which gold standard methodology for assessing sleep and circadian physiology is implemented. We emphasize the importance of assessing both nocturnal and diurnal sleep (naps), valid markers of circadian physiology, and that evaluation of technology is best achieved in protocols in which sleep is mildly disturbed and in populations that are relevant to the intended use-case. We provide details on the design, implementation, challenges, and advantages of this protocol, along with examples of datasets.
Previous studies have led to the beliefs: (1) that short-term memory is best during the night when the body temperature is at its nadir, and (2) that the circadian rhythms of short-term memory and subjective alertness are driven by oscillators independent from each other and from the body temperature cycle. Unfortunately, these conclusions, which would have major implications for understanding the organization of the human circadian timing system, are largely based on field and laboratory studies, which in many cases sampled data infrequently and/or limited data collection to normal waking hours. In order to investigate these points further, we have monitored behavioural variables in two different protocols under controlled laboratory conditions: (1) during a period of 36-60 h of sustained wakefulness; and (2) during forced desynchrony between the body temperature and sleep/wake cycles, allowing testing of non-sleep-deprived subjects at all circadian phases. Contrary to earlier findings, we report here that the circadian rhythm of short-term memory varies in parallel with the circadian rhythms of subjective alertness, calculation performance, and core body temperature under both these experimental conditions. These results challenge the notion that short-term memory is inversely linked to the body temperature cycle and suggest that the human circadian pacemaker, which drives the body temperature cycle, is the primary determinant of endogenous circadian variations in subjective alertness and calculation performance as well as in the immediate recall of meaningful material.
[This corrects the article DOI: 10.2196/46338.].
Background Nocturnal disturbance is frequently observed in dementia and is a major contributor to institutionalisation. Unobtrusive technology that can quantify sleep/wake and determine bed occupancy during the major nocturnal sleep episode may be beneficial for long-term clinical monitoring and the carer. Such technologies have, however, not been validated in older people. Here we assessed the performance of the Withings Sleep Mattress (WSM) in a heterogenous older population to ensure external validity. Method Eighteen participants (65 – 80 years, 10M:8F) completed 7-12 days of sleep/wake monitoring at home prior to an overnight laboratory session. WSM performance was compared to gold-standard (laboratory polysomnography [PSG] with video) and silver standard (actiwatch [AWS] and sleep diary at home). WSM data were downloaded from a third party API and the minute-to-minute sleep/wake timeseries extracted and time-ordered to create a sleep profile. Discontinuities in the timeseries were labelled as ‘missing data’ events. Results Participants contributed 107 nights with WSM and PSG or AWS data. In the laboratory, the overall epoch to epoch agreement (accuracy) of sleep/wake detection of WSM compared to PSG was 0.71 (sensitivity 0.8; specificity 0.45) and to AWS was 0.74 (sensitivity 0.77; specificity 0.53). Visual inspection of video recordings demonstrated that 20 of 21 ‘missing data’ events were true ‘out of bed’ events. These events were always associated with an increase in activity (AWS). At home, all 97 WSM ‘missing data’ events that occurred within the major nocturnal sleep episode defined by sleep diary data, were associated with an increase in activity levels in the AWS data and 36 of these events were also associated with an increase in light levels, indicating that the participant had left the bed. In several participants, data recorded by the WSM during daytime coincided with reported naps in the sleep diary. Conclusion Although WSM cannot reliably distinguish between sleep and wake, the presence/absence of data in WSM seem to be an accurate representation of whether older people are in or out of bed (bed occupancy). Thus, in dementia, this contactless, low-burden technology may be able to provide information about nocturnal disturbances and daytime naps in bed.
The effects of orexinergic peptides are diverse and are mediated by orexin-1 and orexin-2 receptors. Antagonists that target both receptors have been shown to promote sleep initiation and maintenance. Here, we investigated the role of the orexin-2 receptor in sleep regulation in a randomised, double-blind, placebo-controlled, three-period crossover clinical trial using two doses (20 and 50 mg) of a highly selective orexin-2 receptor antagonist (2-SORA) (JNJ-48816274). We used a phase advance model of sleep disruption where sleep initiation is scheduled in the circadian wake maintenance zone. We assessed objective and subjective sleep parameters, pharmacokinetic profiles and residual effects on cognitive performance in 18 healthy male participants without sleep disorders. The phase advance model alone (placebo condition) resulted in disruption of sleep at the beginning of the sleep period compared to baseline sleep (scheduled at habitual time). Compared to placebo, both doses of JNJ-48816274 significantly increased total sleep time, REM sleep duration and sleep efficiency, and reduced latency to persistent sleep, sleep onset latency, and REM latency. All night EEG spectral power density for both NREM and REM sleep were unaffected by either dose. Participants reported significantly better quality of sleep and feeling more refreshed upon awakening following JNJ-48816274 compared to placebo. No significant residual effects on objective performance measures were observed and the compound was well tolerated. In conclusion, the selective orexin-2 receptor antagonist JNJ-48816274 rapidly induced sleep when sleep was scheduled earlier in the circadian cycle and improved self-reported sleep quality without impact on waking performance.
The original article [1] mistakenly swapped the figure images for Figs 17 and 18. This has since been corrected.
Common cold sufferers frequently report sleep disruption during the symptomatic period of infections. We examined the effects of treatment with a topical aromatic pharmaceutical ointment (Vicks VapoRub®), on associated sleep disturbances. The effects of Vicks VapoRub® versus placebo (petrolatum ointment) on subjective and objective measured sleep parameters were assessed in an exploratory study of 100 common cold patients, in a randomized, single blind, controlled, two-arm, parallel design study. The primary efficacy variable was subjective sleep quality measured with the SQSQ (Subjective Quality of Sleep Questionnaire). Additional measures included, ease of falling asleep and depth of sleep (measured with a post-sleep Visual Analog Scale), total sleep time, sleep onset latency, activity score, percentage of sleep, sleep efficiency (measured with actigraphy and SQSQ) and sleep quality index measured with a modified Karolinska Sleep Diary (KSD). The primary endpoint, “How was the quality of your sleep last night?” showed a statistically significant difference in change from baseline in favour of VapoRub treatment (p = 0.0392) versus placebo. Positive effects of VapoRub versus placebo were also observed for “How refreshed did you feel upon waking up?” (p = 0.0122) (SQSQ), “Did you get enough sleep?” (p = 0.0036) (KSD), “How was it to get up?” (p = 0.0120) (KSD) and “Do you feel well-rested?” (p = 0.0125) (KSD). No statistically significant changes from baseline versus placebo were detected in the Actiwatch endpoints. Vicks VapoRub®when applied before retiring to bed can reduce subjective sleep disturbances during a common cold. The results of this exploratory study support the belief among patients that the use of VapoRub improves subjective sleep quality during common cold which was associated with more refreshing sleep.
Circadian organization of the mammalian transcriptome is achieved by rhythmic recruitment of key modifiers of chromatin structure and transcriptional and translational processes. These rhythmic processes, together with posttranslational modification, constitute circadian oscillators in the brain and peripheral tissues, which drive rhythms in physiology and behavior, including the sleep-wake cycle. In humans, sleep is normally timed to occur during the biological night, when body temperature is low and melatonin is synthesized. Desynchrony of sleep-wake timing and other circadian rhythms, such as occurs in shift work and jet lag, is associated with disruption of rhythmicity in physiology and endocrinology. However, to what extent mistimed sleep affects the molecular regulators of circadian rhythmicity remains to be established. Here, we show that mistimed sleep leads to a reduction of rhythmic transcripts in the human blood transcriptome from 6.4% at baseline to 1.0% during forced desynchrony of sleep and centrally driven circadian rhythms. Transcripts affected are key regulators of gene expression, including those associated with chromatin modification (methylases and acetylases), transcription (RNA polymerase II), translation (ribosomal proteins, initiation, and elongation factors), temperature-regulated transcription (cold inducible RNA-binding proteins), and core clock genes including CLOCK and ARNTL (BMAL1). We also estimated the separate contribution of sleep and circadian rhythmicity and found that the sleep-wake cycle coordinates the timing of transcription and translation in particular. The data show that mistimed sleep affects molecular processes at the core of circadian rhythm generation and imply that appropriate timing of sleep contributes significantly to the overall temporal organization of the human transcriptome.
Symbolic dynamic analysis (SDA) methods have been applied to biomedical signals and have been proven efficient in characterising differences in the electroencephalogram (EEG) in various conditions (e.g., epilepsy, Alzheimer’s and Parkinson’s diseases). In this study, we investigated the use of SDA on EEGs recorded during sleep. Lempel-Ziv Complexity (LZC), Permutation Entropy (PE), Permutation Lempel-Ziv Complexity (PLZC), as well as power spectral analysis based on the fast Fourier transform (FFT), were applied to 8-h sleep EEG recordings in healthy men (n=31) and women (n=29), aged 20-74 years. The results of the SDA methods and FFT analysis were compared and the effects of age and sex were investigated. Surrogate data were used to determine whether the findings with SDA methods truly reflected changes in non-linear dynamics of the EEG and not merely changes in the power spectrum. The surrogate data analysis showed that LZC merely reflected spectral changes in EEG activity, whereas PE and PLZC reflected genuine changes in the non-linear dynamics of the EEG. All three SDA techniques distinguished the vigilance states (i.e. wakefulness, REM sleep, NREM sleep and its sub stages: stage 1, stage 2 and slow wave sleep). Complexity of the sleep EEG increased with ageing. Sex on the other hand did not affect the complexity values assessed with any of these three SDA methods, even though FFT detected sex differences. This study shows that SDA provides additional insights into the dynamics of sleep EEG and how it is affected by ageing.
In this study, a novel hybrid tensor factorisation and deep learning approach has been proposed and implemented for sleep pose identification and classification of twelve different sleep postures. We have applied tensor factorisation to infrared (IR) images of 10 subjects to extract group-level data patterns, undertake dimensionality reduction and reduce occlusion for IR images. Pre-trained VGG-19 neural network has been used to predict the sleep poses under the blanket. Finally, we compared our results with those without the factorisation stage and with CNN network. Our new pose detection method outperformed the methods solely based on VGG-19 and 4-layer CNN network. The average accuracy for 10 volunteers increased from 78.1% and 75.4% to 86.0%.
Prolonged wakefulness alters cortical excitability, which is essential for proper brain function and cognition. However, besides prior wakefulness, brain function and cognition are also affected by circadian rhythmicity. Whether the regulation of cognition involves a circadian impact on cortical excitability is unknown. Here, we assessed cortical excitability from scalp EEG-responses to transcranial magnetic stimulation in 22 participants during 29-h of wakefulness under constant conditions. Data reveal robust circadian dynamics of cortical excitability that were strongest in those individuals with highest endocrine markers of circadian amplitude. In addition, the time course of cortical excitability correlated with changes in EEG synchronization and cognitive performance. These results demonstrate that the crucial factor for cortical excitability, and basic brain function in general, is the balance between circadian rhythmicity and sleep need, rather than sleep homeostasis alone. These findings have implications for clinical applications such as noninvasive brain stimulation in neurorehabilitation.
Sleep quality is an important determinant of human health and wellbeing. Novel technologies that can quantify sleep quality at scale are required to enable the diagnosis and epidemiology of poor sleep. One important indicator of sleep quality is body posture. In this paper, we present the design and implementation of a non-contact sleep monitoring system that analyses body posture and movement. Supervised machine learning strategies applied to noncontact vision-based infrared camera data using a transfer learning approach, successfully quantified sleep poses of participants covered by a blanket. This represents the first occasion that such a machine learning approach has been used to successfully detect four predefined poses and the empty bed state during 8-10 hour overnight sleep episodes representing a realistic domestic sleep situation. The methodology was evaluated against manually scored sleep poses and poses estimated using clinical polysomnography measurement technology. In a cohort of 12 healthy participants, we find that a ResNet-152 pre-trained network achieved the best performance compared with the standard de novo CNN network and other pre-trained networks. The performance of our approach was better than other video-based methods for sleep pose estimation and produced higher performance compared to the clinical standard for pose estimation using a polysomnography position sensor. It can be concluded that infrared video capture coupled with deep learning AI can be successfully used to quantify sleep poses as well as the transitions between poses in realistic nocturnal conditions, and that this non-contact approach provides superior pose estimation compared to currently accepted clinical methods.
Introduction: Individual differences in response to sleep loss have been described in various settings including driver sleepiness. A potential biological marker for this differential vulnerability is a PERIOD3 (PER3) Variable Number (4 or 5) Tandem Repeat polymorphism (rs57875989), for which homozygosity for the 5 repeat (PER35/5) has been associated with increased homeostatic sleep pressure and cognitive performance deficits in laboratory conditions. This is the first study so far experimentally investigating the effect of this polymorphism on sleepiness and performance outside the laboratory. Methods: 18 PER3 4/4 homozygotes and 10 PER3 5/5 homozygotes drove during day, evening and night for approximately 90 minutes on real roads. Subjective sleepiness was measured every 5th minute, physiological sleepiness (blink duration, delay of eyelid reopening) was measured continuously. Driving performance was averaged over the whole condition.Statistical analyses were conducted using multilevel mixed effects regression modelling. Results: Subjective sleepiness showed a steeper rise during evening and night conditions in PER3 5/5 individuals. The PER3 polymorphism was also associated with individual differences observed in one of the physiological sleepiness indicators (delay of eyelid reopening). While the standard deviation of lateral position and blink duration showed clear effects of condition and time on task, PER3 genotype was not significantly related to individual differences in these measures. Conclusion: The PER3 VNTR polymorphism contributed significantly to individual differences in subjective and physiological sleepiness during real road driving; yet observed individual differences were still pronounced.
Background Sleep disturbances are both risk factors for and symptoms of dementia. Current methods for assessing sleep disturbances are largely based on either polysomnography (PSG) which is costly and inconvenient, or self‐ or care‐giver reports which are prone to measurement error. Low‐cost methods to monitor sleep disturbances longitudinally and at scale can be useful for assessing symptom development. Here, we develop deep learning models that use multimodal variables (accelerometers and temperature) recorded by the AX3 to accurately identify sleep and wake epochs and derive sleep parameters. Method Eighteen men and women (65‐80y) participated in a sleep laboratory‐based study in which multiple devices for sleep monitoring were evaluated. PSGs were recorded over a 10‐h period and scored according to established criteria per 30 sec epochs. Tri‐axial accelerometers and temperature signals were captured with an Axivity AX3, at 100Hz and 1Hz, respectively, throughout a 19‐h period, including 10‐h concurrent PSG recording and 9‐h of wakefulness. We developed and evaluated a supervised deep learning algorithm to detect sleep and wake epochs and determine sleep parameters from the multimodal AX3 raw data. We validated our results with gold standard PSG measurements and compared our algorithm to the Biobank accelerometer analysis toolbox. Single modality (accelerometer or temperature) and multimodality (both signals) approaches were evaluated using the 3‐fold cross‐validation. Result The proposed deep learning model outperformed baseline models such as the Biobank accelerometer analysis toolbox and conventional machine learning classifiers (Random Forest and Support Vector Machine) by up to 25%. Using multimodal data improved sleep and wake classification performance (up to 18% higher) compared with the single modality. In terms of the sleep parameters, our approach boosted the accuracy of estimations by 11% on average compared to the Biobank accelerometer analysis toolbox. Conclusion In older adults without dementia, combining multimodal data from AX3 with deep learning methods allows satisfactory quantification of sleep and wakefulness. This approach holds promise for monitoring sleep behaviour and deriving accurate sleep parameters objectively and longitudinally from a low‐cost wearable sensor. A limitation of our current study is that the participants were healthy older adults: future work will focus on people living with dementia.
Sleep is considered essential for the brain and body. A predominant concept is that sleep is regulated by circadian rhythmicity and sleep homeostasis, processes that were posited to be functionally and mechanistically separate. Here we review and re-evaluate this concept and its assumptions using findings from recent human and rodent studies. Alterations in genes that are central to circadian rhythmicity affect not only sleep timing but also putative markers of sleep homeostasis such as electroencephalogram slow-wave activity (SWA). Perturbations of sleep change the rhythmicity in the expression of core clock genes in tissues outside the central clock. The dynamics of recovery from sleep loss vary across sleep variables: SWA and immediate early genes show an early response, but the recovery of non-rapid eye movement and rapid eye movement sleep follows slower time courses. Changes in the expression of many genes in response to sleep perturbations outlast the effects on SWA and time spent asleep. These findings are difficult to reconcile with the notion that circadian- and sleep-wake-driven processes are mutually independent and that the dynamics of sleep homeostasis are reflected in a single variable. Further understanding of how both sleep and circadian rhythmicity contribute to the homeostasis of essential physiological variables may benefit from the assessment of multiple sleep and molecular variables over longer time scales.
Sleep disturbance is a prevalent and disabling comorbidity in Parkinson's disease (PD). We performed multi-night (n = 57) at-home intracranial recordings from electrocorticography and subcortical electrodes using sensing-enabled Deep Brain Stimulation (DBS), paired with portable polysomnography in four PD participants and one with cervical dystonia (clinical trial: NCT03582891). Cortico-basal activity in delta increased and in beta decreased during NREM (N2 + N3) versus wakefulness in PD. DBS caused further elevation in cortical delta and decrease in alpha and low-beta compared to DBS OFF state. Our primary outcome demonstrated an inverse interaction between subcortical beta and cortical slow-wave during NREM. Our secondary outcome revealed subcortical beta increases prior to spontaneous awakenings in PD. We classified NREM vs. wakefulness with high accuracy in both traditional (30 s: 92.6 ± 1.7%) and rapid (5 s: 88.3 ± 2.1%) data epochs of intracranial signals. Our findings elucidate sleep neurophysiology and impacts of DBS on sleep in PD informing adaptive DBS for sleep dysfunction.
Study Objectives Assess the validity of a subjective measure of sleepiness as an indicator of sleep drive by quantifying associations between intra-individual variation in evening sleepiness and bedtime, sleep duration, and next morning and subsequent evening sleepiness, in young adults. Methods Sleep timing and sleepiness were assessed in 19 students in late autumn and late spring on a total of 771 days. Karolinska Sleepiness Scales (KSS) were completed at half-hourly intervals at fixed clock times starting four hours prior to participants’ habitual bedtime, and in the morning. Associations between sleepiness and sleep timing were evaluated by mixed model and non-parametric approaches and simulated with a mathematical model for the homeostatic and circadian regulation of sleepiness. Results Intra-individual variation in evening sleepiness was very large, covering four or five points on the 9-point KSS scale, and was significantly associated with subsequent sleep timing. On average, a one point higher KSS value was followed by 20 min earlier bedtime, which led to 11 min longer sleep, which correlated with lower sleepiness next morning and following evening. Associations between sleepiness and sleep timing were stronger in early compared to late sleepers. Model simulations indicated that the directions of associations between sleepiness and sleep timing are in accordance with their homeostatic and circadian regulation, even though much of the variance in evening sleepiness and details of its time course remain unexplained by the model. Conclusion Subjective sleepiness is a valid indicator of the drive for sleep which, if acted upon, can reduce insufficient sleep.
Survival prediction is integral to oncology and palliative care, yet robust prognostic models remain elusive. We assessed the feasibility of combining actigraphy, sleep diary data, and routine clinical parameters to prognosticate. Fifty adult outpatients with advanced cancer and estimated prognosis of
Cognitive performance deteriorates during extended wakefulness and circadian phase misalignment, and some individuals are more affected than others. Whether performance is affected similarly across cognitive domains, or whether cognitive processes involving Executive Functions are more sensitive to sleep and circadian misalignment than Alertness and Sustained Attention, is a matter of debate.
Insufficient sleep and circadian rhythm disruption are associated with negative health outcomes, including obesity, cardiovascular disease, and cognitive impairment, but the mechanisms involved remain largely unexplored. Twenty-six participants were exposed to 1 wk of insufficient sleep (sleep-restriction condition 5.70 h, SEM = 0.03 sleep per 24 h) and 1 wk of sufficient sleep (control condition 8.50 h sleep, SEM = 0.11). Immediately following each condition, 10 whole-blood RNA samples were collected from each participant, while controlling for the effects of light, activity, and food, during a period of total sleep deprivation. Transcriptome analysis revealed that 711 genes were up- or down-regulated by insufficient sleep. Insufficient sleep also reduced the number of genes with a circadian expression profile from 1,855 to 1,481, reduced the circadian amplitude of these genes, and led to an increase in the number of genes that responded to subsequent total sleep deprivation from 122 to 856. Genes affected by insufficient sleep were associated with circadian rhythms (PER1, PER2, PER3, CRY2, CLOCK, NR1D1, NR1D2, RORA, DEC1, CSNK1E), sleep homeostasis (IL6, STAT3, KCNV2, CAMK2D), oxidative stress (PRDX2, PRDX5), and metabolism (SLC2A3, SLC2A5, GHRL, ABCA1). Biological processes affected included chromatin modification, gene-expression regulation, macromolecular metabolism, and inflammatory, immune and stress responses. Thus, insufficient sleep affects the human blood transcriptome, disrupts its circadian regulation, and intensifies the effects of acute total sleep deprivation. The identified biological processes may be involved with the negative effects of sleep loss on health, and highlight the interrelatedness of sleep homeostasis, circadian rhythmicity, and metabolism.
Sleep timing varies between individuals and can be altered in mental and physical health conditions. Sleep and circadian sleep phenotypes, including circadian rhythm sleep-wake disorders, may be driven by endogenous physiological processes, exogeneous environmental light exposure along with social constraints and behavioural factors. Identifying the relative contributions of these driving factors to different phenotypes is essential for the design of personalised interventions. The timing of the human sleep-wake cycle has been modelled as an interaction of a relaxation oscillator (the sleep homeostat), a stable limit cycle oscillator with a near 24-hour period (the circadian process), man-made light exposure and the natural light-dark cycle generated by the Earth's rotation. However, these models have rarely been used to quantitatively describe sleep at the individual level. Here, we present a new Homeostatic-Circadian-Light model (HCL) which is simpler, more transparent and more computationally efficient than other available models and is designed to run using longitudinal sleep and light exposure data from wearable sensors. We carry out a systematic sensitivity analysis for all model parameters and discuss parameter identifiability. We demonstrate that individual sleep phenotypes in each of 34 older participants (65-83y) can be described by feeding individual participant light exposure patterns into the model and fitting two parameters that capture individual average sleep duration and timing. The fitted parameters describe endogenous drivers of sleep phenotypes. We then quantify exogenous drivers using a novel metric which encodes the circadian phase dependence of the response to light. Combining endogenous and exogeneous drivers better explains individual mean mid-sleep (adjusted R-squared 0.64) than either driver on its own (adjusted R-squared 0.08 and 0.17 respectively). Critically, our model and analysis highlights that different people exhibiting the same sleep phenotype may have different driving factors and opens the door to personalised interventions to regularize sleep-wake timing that are readily implementable with current digital health technology.
Objectives: Evidence for a causal relationship between sleep-loss and metabolism is derived primarily from short-term sleep deprivation studies in the laboratory. The objective of this study was to investigate whether small changes in sleep duration over a three week period while participants are living in their normal environment lead to changes in insulin sensitivity and other metabolic parameters. Methods: Nineteen healthy, young, normal-weight men were randomised to either sleep restriction (habitual bedtime minus 1.5 h) or a control condition (habitual bedtime) for three weeks. Weekly assessments of insulin sensitivity by hyperinsulinaemic-euglycaemic clamp, anthropometry, vascular function, leptin and adiponectin were made. Sleep was assessed continuously using actigraphy and diaries. Results: Assessment of sleep by actigraphy confirmed that the intervention reduced daily sleep duration by 01:19 ± 00:15 (SE; p < 0.001). Sleep restriction led to changes in insulin sensitivity, body weight and plasma concentrations of leptin which varied during the three week period. There was no effect on plasma adiponectin or vascular function. Conclusions: Even minor reductions in sleep duration lead to changes in insulin sensitivity, body weight and other metabolic parameters which vary during the exposure period. Larger and longer longitudinal studies of sleep restriction and sleep extension are warranted. © 2013 Elsevier Inc.
Sleep complaints and irregular sleep patterns, such as curtailed sleep during workdays and longer and later sleep during weekends are common. It is often implied that differences in circadian period and in entrained phase contribute to these patterns but few data are available. We assessed parameters of the circadian rhythm of melatonin at baseline and in a forced desynchrony protocol in 35 participants (18 women) with no sleep disorders. Intrinsic circadian period varied between 23h50min and 24h31min and correlated positively (n=31, rs=0.43, P=0.017) with the timing of the melatonin rhythm relative to habitual bedtime. This phase of the melatonin rhythm correlated with the insomnia severity score (n=35, rs=0.47, P=0.004). Self-reported time in bed (TIB) during free days also correlated with the timing of the melatonin rhythm (n=35, rs=0.43, P=0.01) as well as with circadian period (n=31, rs=0.47, P=0.007) such that individuals with a more delayed melatonin rhythm or a longer circadian period reported longer sleep during the weekend. The increase in TIB during the free days correlated positively with circadian period (n=31, rs=0.54, P=0.002). Polysomnographically-assessed latency to persistent sleep (n=34, rs=0.48, P=0.004) correlated with the timing of the melatonin rhythm when participants were sleeping at their habitual bedtimes in the laboratory. This correlation was significantly stronger in women than in men (Z=2.38, P=0.017). The findings show that individual differences in period and phase of the circadian melatonin rhythm associate with differences in sleep and imply that individuals with a long circadian period are at risk of developing sleep problems.
Adequate synchronisation of endogenous circadian rhythms to external time is beneficial for human health [1]. But how circadian time (biological time) and the numbers on the clock (clock time) are related is tricky to understand, as many of us experience when we change from standard time (ST) to daylight saving time (DST) and during jet-lag. How confused we can be is also exemplified by two bills currently making their way through the Californian state legislature. Senate Bill SB-328 Pupil Attendance: School Start Time [2] prohibits middle and high schools from starting earlier than 8:30 in the morning. Senate Bill AB-807 Daylight Saving Time [3] would result in a switch to permanent DST. Similar debates on school start times and DST are happening throughout North America and Europe. Here we explain why a switch to permanent DST could negate any beneficial effects of delaying school start times.
In this study, a novel sleep pose identification method has been proposed for classifying 12 different sleep postures using a two-step deep learning process. For this purpose, transfer learning as an initial stage retrains a well-known CNN network (VGG-19) to categorise the data into four main pose classes, namely: supine, left, right, and prone. According to the decision made by VGG-19, subsets of the image data are next passed to one of four dedicated sub-class CNNs. As a result, the pose estimation label is further refined from one of four sleep pose labels to one of 12 sleep pose labels. 10 participants contributed for recording infrared (IR) images of 12 pre-defined sleep positions. Participants were covered by a blanket to occlude the original pose and present a more realistic sleep situation. Finally, we have compared our results with (1) the traditional CNN learning from scratch and (2) retrained VGG-19 network in one stage. The average accuracy increased from 74.5% & 78.1% to 85.6% compared with (1) & (2) respectively.
Circadian organization of the mammalian transcriptome is achieved by rhythmic recruitment of key modifiers of chromatin structure and transcriptional and translational processes. These rhythmic processes, together with posttranslational modification, constitute circadian oscillators in the brain and peripheral tissues, which drive rhythms in physiology and behavior, including the sleep–wake cycle. In humans, sleep is normally timed to occur during the biological night, when body temperature is low and melatonin is synthesized. Desynchrony of sleep–wake timing and other circadian rhythms, such as occurs in shift work and jet lag, is associated with disruption of rhythmicity in physiology and endocrinology. However, to what extent mistimed sleep affects the molecular regulators of circadian rhythmicity remains to be established. Here, we show that mistimed sleep leads to a reduction of rhythmic transcripts in the human blood transcriptome from 6.4% at baseline to 1.0% during forced desynchrony of sleep and centrally driven circadian rhythms. Transcripts affected are key regulators of gene expression, including those associated with chromatin modification (methylases and acetylases), transcription (RNA polymerase II), translation (ribosomal proteins, initiation, and elongation factors), temperature-regulated transcription (cold inducible RNA-binding proteins), and core clock genes including CLOCK and ARNTL (BMAL1). We also estimated the separate contribution of sleep and circadian rhythmicity and found that the sleep–wake cycle coordinates the timing of transcription and translation in particular. The data show that mistimed sleep affects molecular processes at the core of circadian rhythm generation and imply that appropriate timing of sleep contributes significantly to the overall temporal organization of the human transcriptome.
Electroencephalography (EEG) recordings represent a vital component of the assessment of sleep physiology, but the methodology presently used is costly, intrusive to participants, and laborious in application. There is a recognized need to develop more easily applicable yet reliable EEG systems that allow unobtrusive long-term recording of sleep-wake EEG ideally away from the laboratory setting. cEEGrid is a recently developed flex-printed around-the-ear electrode array, which holds great potential for sleep-wake monitoring research. It is comfortable to wear, simple to apply, and minimally intrusive during sleep. Moreover, it can be combined with a smartphone-controlled miniaturized amplifier and is fully portable. Evaluation of cEEGrid as a motion-tolerant device is ongoing, but initial findings clearly indicate that it is very well suited for cognitive research. The present study aimed to explore the suitability of cEEGrid for sleep research, by testing whether cEEGrid data affords the signal quality and characteristics necessary for sleep stage scoring. In an accredited sleep laboratory, sleep data from cEEGrid and a standard PSG system were acquired simultaneously. Twenty participants were recorded for one extended nocturnal sleep opportunity. Fifteen data sets were scored manually. Sleep parameters relating to sleep maintenance and sleep architecture were then extracted and statistically assessed for signal quality and concordance. The findings suggest that the cEEGrid system is a viable and robust recording tool to capture sleep and wake EEG. Further research is needed to fully determine the suitability of cEEGrid for basic and applied research as well as sleep medicine.
Symbolic dynamic analysis (SDA) methods have been applied to biomedical signals and have been proven efficient in characterising differences in the electroencephalogram (EEG) in various conditions (e.g., epilepsy, Alzheimer’s, and Parkinson’s diseases). In this study, we investigated the use of SDA on EEGs recorded during sleep. Lempel-Ziv complexity (LZC), permutation entropy (PE), and permutation Lempel-Ziv complexity (PLZC), as well as power spectral analysis based on the fast Fourier transform (FFT), were applied to 8-h sleep EEG recordings in healthy men (n=31) and women (n=29), aged 20-74 years. The results of the SDA methods and FFT analysis were compared and the effects of age and sex were investigated. Surrogate data were used to determine whether the findings with SDA methods truly reflected changes in nonlinear dynamics of the EEG and not merely changes in the power spectrum. The surrogate data analysis showed that LZC merely reflected spectral changes in EEG activity, whereas PE and PLZC reflected genuine changes in the nonlinear dynamics of the EEG. All three SDA techniques distinguished the vigilance states (i.e., wakefulness, REM sleep, NREM sleep, and its sub-stages: stage 1, stage 2, and slow wave sleep). Complexity of the sleep EEG increased with ageing. Sex on the other hand did not affect the complexity values assessed with any of these three SDA methods, even though FFT detected sex differences. This study shows that SDA provides additional insights into the dynamics of sleep EEG and how it is affected by ageing.
Genome biology approaches have made enormous contributions to our understanding of biological rhythms, particularly in identifying outputs of the clock, including RNAs, proteins, and metabolites, whose abundance oscillates throughout the day. These methods hold significant promise for future discovery, particularly when combined with computational modeling. However, genome-scale experiments are costly and laborious, yielding “big data” that are conceptually and statistically difficult to analyze. There is no obvious consensus regarding design or analysis. Here we discuss the relevant technical considerations to generate reproducible, statistically sound, and broadly useful genome-scale data. Rather than suggest a set of rigid rules, we aim to codify principles by which investigators, reviewers, and readers of the primary literature can evaluate the suitability of different experimental designs for measuring different aspects of biological rhythms. We introduce CircaInSilico, a web-based application for generating synthetic genome biology data to benchmark statistical methods for studying biological rhythms. Finally, we discuss several unmet analytical needs, including applications to clinical medicine, and suggest productive avenues to address them.
Sleep homeostasis and circadian rhythmicity interact to determine the timing of behavioral activity. Circadian clock genes contribute to circadian rhythmicity centrally and in the periphery, but some also have roles within sleep regulation. The clock gene Period3 (Per3) has a redundant function within the circadian system and is associated with sleep homeostasis in humans. This study investigated the role of PER3 in sleep/wake activity and sleep homeostasis in mice by recording wheel running activity under baseline conditions in wild-type (WT; n = 54) and in PER3-deficient (Per3(-/-); n = 53) mice, as well as EEG-assessed sleep before and after 6 hours of sleep deprivation in WT (n = 7) and Per3(-/-) (n = 8) mice. Whereas total activity and vigilance states did not differ between the genotypes, the temporal distribution of wheel running activity, vigilance states, and EEG delta activity was affected by genotype. In Per3(-/-) mice, running wheel activity was increased and REM sleep and NREM sleep were reduced in the middle of the dark phase, and delta activity was enhanced at the end of the dark phase. At the beginning of the baseline light period, there was less wakefulness and more REM and NREM in Per3(-/-) mice. Per3(-/-) mice spent less time in wakefulness and more time in NREM sleep in the light period immediately after sleep deprivation and REM sleep accumulated more slowly during the recovery dark phase. These data confirm a role for PER3 in sleep/wake timing and sleep homeostasis.
Several neuropsychiatric and neurological disorders have recently been characterized as dysfunctions arising from a ‘final common pathway’ of imbalanced excitation to inhibition within cortical networks. How the regulation of a cortical E/I ratio is affected by sleep and the circadian rhythm however, remains to be established. Here we addressed this issue through the analyses of TMS-evoked responses recorded over a 29h sleep deprivation protocol conducted in young and healthy volunteers. Spectral analyses of TMS-evoked responses in frontal cortex revealed non-linear changes in gamma band evoked oscillations, compatible with an influence of circadian timing on inhibitory interneuron activity. In silico inferences of cell-to-cell excitatory and inhibitory connectivity and GABA/Glutamate receptor time constant based on neural mass modeling within the Dynamic causal modeling framework, further suggested excitation/inhibition balance was under a strong circadian influence. These results indicate that circadian changes in EEG spectral properties, in measure of excitatory/inhibitory connectivity and in GABA/glutamate receptor function could support the maintenance of cognitive performance during a normal waking day, but also during overnight wakefulness. More generally, these findings demonstrate a slow daily regulation of cortical excitation/inhibition balance, which depends on circadian-timing and prior sleep-wake history.
Objectives: Previously, we reported a light-dependant phenotype incircadian regulation in PER3 knockout (Per3-/-) mice. These mice also showed altered sleep architecture and elevated activity levels inthe second half of the dark period. In humans, a polymorphism inPER3 has been associated with diurnal preference, sleep homeo-stasis, and cognitive decline in response to sleep loss. We generated humanised knock-in (KI) mice expressing two variants of the human polymorphism and investigated activity patterns in response to different photoperiods. We also further investigated gene expression profiles of Per3-/- and KI mice during an ultradian light exposure paradigm. Methods: Male and female C57Bl/6 mice, expressing either the 4- or 5- repeat of the human variable number tandem repeat in PER3 (Per34/4 and Per35/5) were exposed to short (8 h), intermediate (12 h) and long (16 h) photoperiods, as well as constant light.Transitions between the conditions were mixed between animals,such that the response to a new photoperiod could be analysed,taking into account different light-histories. Behavioural activity was recorded as running wheel revolutions. In addition, we subjected Per3-/- and KI mice to an ultradian light-dark cycle (3.5 h L–3.5 h D) and analysed whole genome RNA expression at CT 16, in an ultradian light episode. Results: Significant differences between male and female activity were seen. Female mice showed more activity in the second half of the dark period, and overall 24-h activity levels were more than 1.5-fold higher in females. These differences were seen in all genotypes. In constant darkness, both male and female Per34/4 mice showed increased activity in the second half of the dark period, compared toWT and Per35/5 mice. The behavioural responses to photoperiods were diverse, with KI mice appearing to adjust more rapidly to a new photoperiod. Whole genome RNA expression in Per3-/-and KI mice was altered compared to WT mice, and similar pathways were affected in both Per3-/-and KI mice. Conclusion: Here we show behavioural data on a novel humanised mouse model of PER3. In mice, this polymorphism associates with altered activity, especially in the transition between photic conditions.We also observed a consistent difference between male and female activity. This emphasizes the need to not only use transgenic mice but also to include both sexes in animal models of human conditions.
Diagnosis and treatment of circadian rhythm sleep-wake disorders requires assessment of circadian phase of the brain’s circadian pacemaker. The gold-standard univariate method is based on collection of a 24 h time series of plasma melatonin, a suprachiasmatic nucleus driven pineal hormone. We developed and validated a multivariate whole-blood mRNA based predictor of melatonin phase which requires few samples. Transcriptome data were collected under normal, sleep-deprivation and abnormal sleep-timing conditions to assess robustness of the predictor. Partial least square regression (PLSR), applied to the transcriptome, identified a set of 100 biomarkers primarily related to glucocorticoid signaling and immune function. Validation showed that PLSR-based predictors outperform published blood-derived circadian phase predictors. When given one sample as input, the R2 of predicted vs observed phase was 0.74, whereas for two samples taken 12 h apart, R2 was 0.90. This blood transcriptome based model enables assessment of circadian phase from a few samples.
Why do we go to sleep late and struggle to wake up on time? Historically, light-dark cycles were dictated by the solar day, but now humans can extend light exposure by switching on artificial lights. We use a mathematical model incorporating effects of light, circadian rhythmicity and sleep homeostasis to provide a quantitative theoretical framework to understand effects of modern patterns of light consumption on the human circadian system. The model shows that without artificial light humans wake-up at dawn. Artificial light delays circadian rhythmicity and preferred sleep timing and compromises synchronisation to the solar day when wake-times are not enforced. When wake-times are enforced by social constraints, such as work or school, artificial light induces a mismatch between sleep timing and circadian rhythmicity (‘social jet-lag’). The model implies that developmental changes in sleep homeostasis and circadian amplitude make adolescents particularly sensitive to effects of light consumption. The model predicts that ameliorating social jet-lag is more effectively achieved by reducing evening light consumption than by delaying social constraints, particularly in individuals with slow circadian clocks or when imposed wake-times occur after sunrise. These theory-informed predictions may aid design of interventions to prevent and treat circadian rhythm-sleep disorders and social jet-lag.
Polymorphisms in the human circadian clock gene PERIOD3 (PER3) are associated with a wide variety of phenotypes such as diurnal preference, delayed sleep phase disorder, sleep homeostasis, cognitive performance, bipolar disorder, type 2 diabetes, cardiac regulation, cancer, light sensitivity, hormone and cytokine secretion, and addiction. However, the molecular mechanisms underlying these phenotypic associations remain unknown. Per3 knockout mice (Per3
Background and purpose: In work with chronic stroke patients the authors observed that patients frequently appear sleepy and often comment on their poor sleep. Sleep difficulties are frequently reported and indeed clinically recognized in the acute phase post-stroke, but little is known about the sleep and daytime sleepiness of chronic stroke patients with sustained disabilities. The latter, however, deserves clarification because sleep is a critical modulator of health, daytime performance and wellbeing. The present study therefore explored the sleep and sleepiness in a chronic stroke population with sustained physical deficits. Methods: An opportunity sample of 20 patients with chronic low-functioning hemiplegia (12 months) completed the Epworth Sleepiness Scale, Pittsburgh Sleep Quality Index, Medical Outcome Study Short Form 36 and Hospital Anxiety and Depression Scale. Results: Compared to a normative healthy population, long-term stroke survivors reported poorer sleep and greater daytime sleepiness. Increased levels of sleepiness were associated with longer chronicity, whereas nocturnal sleep parameters were not. Conclusions: In line with clinical observations, stroke survivors with sustained physical disabilities report poorer sleep and experience greater levels of sleepiness. Further research in a larger cohort and including objective sleep measures is necessary to investigate the nature and scale of sleep difficulties and daytime sleepiness in more detail so that care and treatment strategies can be developed in due course.
In humans, a primate-specific variable-number tandem-repeat (VNTR) polymorphism (4 or 5 repeats 54 nt in length) in the circadian gene PER3 is associated with differences in sleep timing and homeostatic responses to sleep loss. We investigated the effects of this polymorphism on circadian rhythmicity and sleep homeostasis by introducing the polymorphism into mice and assessing circadian and sleep parameters at baseline and during and after 12 h of sleep deprivation (SD). Microarray analysis was used to measure hypothalamic and cortical gene expression. Circadian behavior and sleep were normal at baseline. The response to SD of 2 electrophysiological markers of sleep homeostasis, electroencephalography (EEG) θ power during wakefulness and δ power during sleep, were greater in the Per3(5/5) mice. During recovery, the Per3(5/5) mice fully compensated for the SD-induced deficit in δ power, but the Per3(4/4) and wild-type mice did not. Sleep homeostasis-related transcripts (e.g., Homer1, Ptgs2, and Kcna2) were differentially expressed between the humanized mice, but circadian clock genes were not. These data are in accordance with the hypothesis derived from human data that the PER3 VNTR polymorphism modifies the sleep homeostatic response without significantly influencing circadian parameters.-Hasan, S., van der Veen, D. R., Winsky-Sommerer, R., Hogben, A., Laing, E. E., Koentgen, F., Dijk, D.-J., Archer, S. N. A human sleep homeostasis phenotype in mice expressing a primate-specific PER3 variable-number tandem-repeat coding-region polymorphism.
Background: Sleep is one of the most important recovery mechanisms available to humans, allowing for recovery from daily strains, and therefore a prerequisite for health. Many workers complain that they are unable to get to sleep at night, and report poor sleep maintenance due to unwanted, ruminative thoughts and concerns about work-related issues. The present study investigated the effects of ruminative thinking on sleep, using self-reported diaries. Method: One-hundred and seven school teachers were asked to keep a diary record of their thoughts about work over a workday evening and were monitored hourly from 17.00 hrs until bedtime. Each individual also completed a diary assessment of their sleep patterns over the same night. Using information obtained from the diaries the sample was divided into high ruminators (those who thought about work issues a lot at bedtime) and low ruminators (those who thought about work issues little at bedtime) using tertile splits. Only individuals who did not work in the hour before bedtime were included in the analysis. Results: Logistic regression analysis revealed (after adjusting for age and gender), that high compared to low ruminators were: 3.5 time more likely to report 'difficulty falling asleep', 4.7 time more likely to report 'difficulty waking up', 5.7 times more likely to report 'difficulty getting back to sleep if awoken during the night', 6.8 times more likely to report 'restless sleep' and 3.4 times more likely to 'feel unrefreshed after awaking'. Relative to the low ruminators, high ruminators also reported that they had thought about work related issues - while trying to fall asleep (p < 0.001), and in the morning before they got out of bed (p < 0.5). Conclusion: Ruminating about work-related issues appears to be associated with self-reported sleep disturbance. It is important therefore that individuals learn to 'switch-off' from work during the evening in order to obtain good quality sleep.
To investigate the effects of subcutaneous detemir on glucose flux, lipid metabolism and brain function, twelve people with type 1 diabetes received in random order 0.5Units/kgBW detemir or NPH insulin. Glucose concentration was clamped at 5mmol/L then increased to 10mmol/L. Glucose production rate (glucose Ra), glucose uptake (glucose Rd) and glycerol production (glycerol Ra) were measured with a constant iv infusion of [6,6(2) H2 ]glucose and [(2) H5 ]glycerol. Electroencephalography direct (DC) and alternating (AC) current potentials were measured. While detemir induced comparable effects on glucose Ra, glucose Rd and glycerol Ra during euglycaemia, compared with NPH, it triggered a distinct negative shift in DC-potentials, with significant treatment effect in frontal cerebrocortical channels (p
Objective/Background The aim of this study was to examine the relationship between overnight consolidation of implicit statistical learning with spindle frequency EEG activity and slow frequency delta power during non-rapid eye movement (NREM) sleep in obstructive sleep apnea (OSA). Patients/Methods Forty-seven OSA participants completed the experiment. Prior to sleep, participants performed a reaction time cover task containing hidden patterns of pictures, about which participants were not informed. After the familiarisation phase, participants underwent overnight polysomnography. 24 h after the familiarisation phase, participants performed a test phase to assess their learning of the hidden patterns, expressed as a percentage of the number of correctly identified patterns. Spindle frequency activity (SFA) and delta power (0.5–4.5 Hz), were quantified from NREM electroencephalography. Associations between statistical learning and sleep EEG, and OSA severity measures were examined. Results SFA in NREM sleep in frontal and central brain regions was positively correlated with statistical learning scores (r = 0.41 to 0.31, p = 0.006 to 0.044). In multiple regression, greater SFA and longer sleep onset latency were significant predictors of better statistical learning performance. Delta power and OSA severity were not significantly correlated with statistical learning. Conclusions These findings suggest spindle activity may serve as a marker of statistical learning capability in OSA. This work provides novel insight into how altered sleep physiology relates to consolidation of implicitly learnt information in patients with moderate to severe OSA.
The human sleep-wake cycle is generated by a circadian process, originating from the suprachiasmatic nuclei, in interaction with a separate oscillatory process: the sleep homeostat. The sleep-wake cycle is normally timed to occur at a specific phase relative to the external cycle of light-dark exposure. It is also timed at a specific phase relative to internal circadian rhythms, such as the pineal melatonin rhythm, the circadian sleep-wake propensity rhythm, and the rhythm of responsiveness of the circadian pacemaker to light. Variations in these internal and external phase relationships, such as those that occur in blindness, aging, morning and evening, and advanced and delayed sleep-phase syndrome, lead to sleep disruptions and complaints. Changes in ocular circadian photoreception, interindividual variation in the near-24-h intrinsic period of the circadian pacemaker, and sleep homeostasis can contribute to variations in external and internal phase. Recent findings on the physiological and molecular-genetic correlates of circadian sleep disorders suggest that the timing of the sleep-wake cycle and circadian rhythms is closely integrated but is, in part, regulated differentially.
The human sleep-wake cycle is generated by a circadian process, originating from the suprachiasmatic nuclei, in interaction with a separate oscillatory process: the sleep homeostat. The sleep-wake cycle is normally timed to occur at a specific phase relative to the external cycle of light-dark exposure. It is also timed at a specific phase relative to interal circadian rhythms, such as the pineal melatonin rhythm, the circadian sleep-wake propensity rhythm, and the rhythm of responsiveness of the circadian pacemaker to light. Variations in these internal and external phase relationships, such as those that occur in blindness, aging, morning and evening, and advanced and delayed sleep-phase syndrome, lead to sleep disruptions and complaints. Changes in ocular circadian photoreception, interindividual variation in the near-24-h intrinsic period of the circadian pacemaker, and sleep homeostasis can contribute to variations in external and internal phase. Recent findings on the physiological and molecular-genetic correlates of circadian sleep disorders suggest that the timing of the sleep-wake cycle and circadian rhythms is closely integrated but is, in part, regulated differentially.
Circadian and other physiological rhythms play a key role in both normal homeostasis and disease processes. Such is the case of circadian and infradian seizure patterns observed in epilepsy. However, these rhythms are not fully exploited in the design of active implantable medical devices. In this paper we explore a new implantable stimulator that implements chronotherapy as a feedforward input to supplement both open-loop and closed-loop methods. This integrated algorithm allows for stimulation to be adjusted to the ultradian, circadian and infradian patterns observed in patients through slowly-varying temporal adjustments of stimulation and algorithm sub-components, while also enabling adaption of stimulation based on immediate physiological needs such as a breakthrough seizure or change of posture. Embedded physiological sensors in the stimulator can be used to refine the baseline stimulation circadian pattern as a “digital zeitgeber,” i.e., a source of stimulus that entrains or synchronizes the subject's natural rhythms. This algorithmic approach is tested on a canine with severe drug-resistant idiopathic generalized epilepsy exhibiting a characteristic diurnal pattern correlated with sleep-wake cycles. Prior to implantation, the canine's cluster seizures evolved to status epilepticus (SE) and required emergency pharmacological intervention. The cranially-mounted system was fully-implanted bilaterally into the centromedian nucleus of the thalamus. Using combinations of time-based modulation, thalamocortical rhythm-specific tuning of frequency parameters as well as fast-adaptive modes based on activity, the canine experienced no further SE events post-implant as of the time of writing (7 months). Importantly, no significant cluster seizures have been observed either, allowing the reduction of rescue medication. The use of digitally-enabled chronotherapy as a feedforward signal to augment adaptive neurostimulators could prove a useful algorithmic method in conditions where sensitivity to temporal patterns are characteristics of the disease state, providing a novel mechanism for tailoring a more patient-specific therapy approach.
Sleep deficiencies and associated performance decrements are common among astronauts during spaceflight missions. Previously, sleep in space was analyzed with a focus on global measures while the intricate structure of sleep oscillations remains largely unexplored. This study extends previous findings by analyzing how spaceflight affects characteristics of sleep spindles and slow waves, two sleep oscillations associated with sleep quality and quantity, in four astronauts before, during and after two Space Shuttle missions. Analysis of these oscillations revealed significantly increased fast spindle density, elevated slow spindle frequency, and decreased slow wave amplitude in space compared to on Earth. These results reflect sleep characteristics during spaceflight on a finer electrophysiological scale and provide an opportunity for further research on sleep in space.
Purpose Using quantitative EEG (qEEG) analysis, we investigated sleep EEG microstructure as correlates of neurobehavioural performance after 24 h of extended wakefulness in untreated OSA. Methods Eight male OSA patients underwent overnight polysomnography (PSG) at baseline followed by 40 h awake with repeated performance testing (psychomotor vigilance task [PVT] and AusEd driving simulator). EEG slowing during REM and spindle density during NREM sleep were calculated using power spectral analysis and a spindle detection algorithm at frontal and central electrode sites. Correlations between sleep EEG microstructure measures and performance after 24-h awake were assessed. Results Greater EEG slowing during REM sleep was associated with slower PVT reaction times (rho = − 0.79, p = 0.02), more PVT lapses (rho = 0.87, p = 0.005) and more AusEd crashes (rho = 0.73, p = 0.04). Decreased spindle density in NREM sleep was also associated with slower PVT reaction times (rho = 0.89, p = 0.007). Traditional PSG measures of disease severity were not consistent correlates of neurobehavioural performance in OSA. Conclusions Sleep EEG microstructure measures recorded during routine PSG are associated with impaired vigilance in OSA patients after sleep deprivation. Significance Quantitative brain oscillatory (or EEG)–based measures of sleep may better reflect the deleterious effects of untreated OSA than traditional PSG metrics in at-risk individuals. Trial Registration ACTRN12606000066583
Background Sleep problems are prevalent in schizophrenia spectrum disorders, and include psychophysiological insomnia, and sleep disturbances caused predominantly by circadian dysregulation. Studies using motion sensing wearables (including actigraphy) have shown a diverse range of rest activity patterns, including mis-timed sleep, irregular sleep, or non-24hr (free-running) sleep patterns. Sleep and circadian rhythm disturbances impact negatively on functioning and quality of life, and long term poor sleep is linked to weight gain, and other serious physical health conditions. Many of the factors supposed to cause or maintain poor sleep in this group are modifiable through behavioural and environmental alterations. Methods An expert consensus study (Delphi study) was conducted to examine and explore the views of relevant experts, regarding the appropriate contents and format of an occupational therapy intervention to improve sleep in schizophrenia spectrum disorders. Experts were recruited with expertise in: occupational therapy, sleep, insomnia, circadian rhythm, and schizophrenia spectrum disorders. Experts with clinical and/or research expertise were sought, as well as service users and carers. We conducted three rounds of online surveys, individual interviews, and a day of content presentation, comments and voting, and discussion groups with service users and carers. Quantitative data were analysed using descriptive statistics and presented graphically for comparisons. Qualitative data (survey comments and interviews) was analysed using thematic analysis. Analysis and data collection was iterative; with later rounds exploring views raised during earlier rounds. The results informed the development of the intervention now being tested in a single group feasibility study recruiting twenty participants. Using mixed methods, this study will evaluate acceptability, homework adherence and trial related procedures, informing adjustments prior to larger scale testing. Results The Delphi study recruited and retained participants well (n=82 of target=85, 66% uptake from professionals approached, 97% completion), suggesting a high level of interest in this topic. Participants reached consensus regarding relevant content within the initial assessment, increasing daytime natural light exposure (when possible), approaches to evening routine, and the approach to activity and occupation. Views varied regarding the relative importance of sleep restriction and building ‘sleep drive’, versus reducing arousal such as through relaxation, mindfulness, or cognitive approaches to anxiety. Often service users’ and carers’ views reflected those of professionals, however on some areas views between these groups differed, such as acceptable levels of intervention burden, and regarding wearables. Overall views highlighted the importance of a tailored intervention, with adjustable and selectable elements. The tailoring of recommendations will be achieved through a thorough assessment, including longitudinal use of light and movement sensing wearables, and a detailed initial interview. Many intervention components are optional based on needs, and the homework recommendations from ‘core’ intervention elements will vary depending upon individual needs, preferences, and baseline problems and situation. Discussion The diversity of sleep disturbance presentations within this group poses a unique challenge when developing a brief first-line intervention. Individually tailored recommendations can be derived through the combination of wearable technology and self-report, and a growing body of work informing models of sleep disturbance in this group.
Sleep is a very rich phenotype and many aspects of sleep differ considerably in the population of healthy individuals (even when only a very narrow age range is considered). Interindividual variation in sleep timing (diurnal preference), sleep duration, sleep structure and the EEG in NREM sleep, and REM sleep and wakefulness, have all been shown to have a genetic basis. The response to challenges of sleep regulatory processes such as sleep deprivation and circadian misalignment has also been shown to vary between individuals. Some of the polymorphic variations in genes contributing to variation in sleep characteristics have now been identified. The most consistent variants include genes associated with circadian clock mechanisms (e.g., CLOCK, PER1-3, BHLHE41), adenosinergic (ADA) and monoaminergic neurotransmission (e.g., DAT1, DRD2, ADRB1), as well as transcription regulating pathways (e.g., PAX8, VRK2, MEIS1). For some of these genes, so far only associations with one aspect of sleep have been reported, for example, HCRTR2 and sleep timing. Variations in other genes have been shown to affect multiple aspects of sleep and wakefulness, as well as the response to sleep loss or pharmacological interventions. For example, PER3, ADA, and BDNF affect the EEG and performance during prolonged waking, whereas DAT1, DRD2, and ADORA2A modulate EEG and response to stimulants such as caffeine and modafinil. Apart from a few genes identified in familial pedigrees with extreme phenotypes (e.g., NPSR1 in a family with natural short sleep), the currently known polymorphic variations explain only a small part of the variation in healthy human sleep phenotypes. Deep phenotyping approaches covering the EEG in all three vigilance states, sleep structure and homeostatic aspects of sleep-wake regulation will be needed to provide further insights into intertwined and widespread consequences of genetic variants for healthy human sleep.
Background Sleep disruption is a common precursor to deterioration and relapse in people living with psychotic disorders. Understanding the temporal relationship between sleep and psychopathology is important for identifying and developing interventions which target key variables that contribute to relapse. Methods We used a purpose-built digital platform to sample self-reported sleep and psychopathology variables over 1 year, in 36 individuals with schizophrenia. Once-daily measures of sleep duration and sleep quality, and fluctuations in psychopathology (positive and negative affect, cognition and psychotic symptoms) were captured. We examined the temporal relationship between these variables using the Differential Time-Varying Effect (DTVEM) hybrid exploratory-confirmatory model. Results Poorer sleep quality and shorter sleep duration maximally predicted deterioration in psychosis symptoms over the subsequent 1–8 and 1–12 days, respectively. These relationships were also mediated by negative affect and cognitive symptoms. Psychopathology variables also predicted sleep quality, but not sleep duration, and the effect sizes were smaller and of shorter lag duration. Conclusions Reduced sleep duration and poorer sleep quality anticipate the exacerbation of psychotic symptoms by approximately 1–2 weeks, and negative affect and cognitive symptoms mediate this relationship. We also observed a reciprocal relationship that was of shorter duration and smaller magnitude. Sleep disturbance may play a causal role in symptom exacerbation and relapse, and represents an important and tractable target for intervention. It warrants greater attention as an early warning sign of deterioration, and low-burden, user-friendly digital tools may play a role in its early detection.
Sleep homeostasis and circadian rhythmicity are two main processes governing both sleep propensity and sleep structure. Sleep homeostasis refers to the processes monitoring how long one has been awake, how long and how deep one has slept, and what sort of sleep one has obtained. Sleep-homeostatic processes ensure that on average one obtains sufficient sleep to restore those functions and variables, which are affected by wakefulness. Circadian rhythmicity, in the context of sleep regulation, refers to the near 24-h variation in the propensity to sleep. Circadian rhythmicity ensures that sleep occurs primarily at the appropriate time of day, which is during the nighttime in humans.
Background Disturbances of sleep and night‐time behaviours are amongst the most disabling symptoms of dementia. They often increase carers’ burden and the risk of institutionalization. The causes are complex and are difficult to investigate because of a lack of acceptable methods for monitoring behaviours in the home. Here we show that a passive under‐mattress can be used to track changes in night‐time behaviour and physiology, and that a range of digital biomarkers produced are informative in understanding the effects of medication changes, disease progression and intercurrent illness in patients living with dementia (PLWD). Method We used contactless Withings Sleep Mattress (WSM) to monitor bed‐occupancy in 4 PLWD (age 74‐93, 3males) enrolled into the CR&T MINDER cohort study. Each participant was tracked over 1000 nights between 2019 and 2021. Minute‐to‐minute timeseries were extracted from WSM to calculate bed occupancy metrics and nocturnal physiology measures (heart and breathing rates (HR/BR)). Raw measures were standardized within subjects by comparing each time point to the mean of the time points that preceded it. We then investigated the relationship between these metrics and clinical events such as infections and medication. Result The 4 case studies illustrate the potential of this technology for passive health monitoring in PLWD. High levels of intraindividual variability in behavioural and physiological metrics were observed. Progressive changes in bed occupancy were observed in two patients with frontotemporal dementia and Alzheimer’s disease (Cases 1&2). Intercurrent illness and medications changes influenced the measures. For example, Patient 1 showed progressive night‐time wandering with increasing time spent out of bed, which improved following the initiation of risperidone. Case 2 showed recurrent episodes of heart failure accompanied by increased nocturnal HR. Cases 3 and 4 showed urinary tract infections, which were accompanied by transient changes in bed occupancy measures. Conclusion Passive monitoring of night‐time behaviour and physiology is a simple and cost‐effective way to monitor the effects of medication change and medical events in PLWD. Digital biomarkers derived from these simple measures hold great promise as measures of dementia progression, to enhance dementia care by identifying at risk individuals and as novel clinical outcome measures in clinical trials.
© Cambridge University Press 2013.It would be extremely unusual, not to mention highly inconvenient, if everyone woke up and went about their daily routines at the same time. Fortunately this is not the case, and humans display a wide range of sleep–wake timing preferences. Some of us like to wake up and get things done in the morning (so-called larks, or morning types), others prefer to be active later in the day and night (owls, or evening types), and many are in between or a mixture of the two. The range in sleep–wake timing is considerable and differences in preferred bedtime and wake time can be as much as 2–3 The on average between morning and evening types [1], and in circadian rhythm sleep phase disorders, bedtimes can range from 7–9 p.m. (advanced) to 2–6 a.m. (delayed) [2]. It has often been assumed that diurnal preference (morningness versus eveningness) is not an acquired characteristic but relates to biological factors involved in the circadian timing system that regulates the optimum times for waking performance and sleep–wake timing. However, current understanding of factors influencing variation in sleep–wake timing and optimal timing of waking performance emphasizes the interactive contribution of social factors, such as work schedules and leisure time, and biological factors. Underlying biological factors include the timing (phase of entrainment) of the endogenous circadian rhythmicity relative to clock time, and the light–dark cycle [3]. The phase of entrainment is determined by the intrinsic period of the circadian clock, as well as sensitivity to the effects of light on the circadian clock. In addition, sleep homeostatic mechanisms also play an important role in sleep–wake timing. This implies that diurnal preference could be related to any of these three main factors: circadian period, light sensitivity, and sleep homeostasis.
Background: Sleep and circadian rhythm disturbances in schizophrenia are common, but incompletely characterised. We aimed to describe and compare the magnitude and heterogeneity of sleep-circadian alterations in remitted schizophrenia, and compare them with those in inter-episode bipolar disorder. Methods: EMBASE, Medline, and PsycINFO were searched for case-control studies reporting actigraphic parameters in remitted schizophrenia or bipolar disorder. Standardised and absolute mean differences between patients and controls were quantified using Hedges-g, and patient-control differences in variability were quantified using the mean-scaled coefficient of variation ratio (CVR). A wald-type test compared effect-sizes between disorders. Results: Thirty studies reporting on 967 patients and 803 controls were included. Compared to controls, both schizophrenia and bipolar groups had significantly longer total sleep time (mean difference (minutes) [95%CI] = 99.9 [66.8, 133.1] and 31.1 [19.3, 42.9], respectively), time in bed (MD = 77.8 [13.7, 142.0] and 50.3 [20.3, 80.3]), but also greater sleep latency (16.5 [6.1, 27.0] and 2.6 [0.5, 4.6]) and reduced motor activity (SMD [95%CI] = -0.86 [-1.22, -0.51] and -0.75 [-1.20, -0.29]). Effect sizes were significantly greater in schizophrenia compared to the bipolar disorder group for total sleep time, sleep latency, and wake after sleep onset. CVR was significantly elevated in both diagnoses for total sleep time, time in bed, and relative amplitude. Conclusions: In both disorders, longer overall sleep duration, but also disturbed initiation, continuity and reduced motor activity were found. Common, modifiable factors may be associated with these sleep-circadian phenotypes, and advocate for further development of transdiagnostic interventions that target them.
© 2012 by Oxford University Press, Inc. All rights reserved.The alternation of sleep and wakefulness is a major determinant of the structure and quality of our lives. The sleep- wake cycle is regulated by a fine-tuned balance between two physiological processes: sleep homeostasis, which measures sleep debt, and circadian rhythmicity, which determines the optimal internal (biological) time for sleep and wakefulness. Sleep homeostasis and circadian rhythmicity together influence many aspects of sleep, such as the time it takes to fall asleep, the timing of awakening and the interruptions of sleep, as well as the duration of rapid eye movement sleep, slow-wave sleep and specific brainwaves during sleep, such as sleep spindles. Alterations in the balance between sleep homeostasis and circadian rhythmicity contribute to sleep phenotypes such as morningness-eveningness and short-long sleepers as well as sleep disturbances. Emerging insights into the environmental, behavioral, physiological, neurochemical and molecular-genetic determinants of sleep homeostasis and circadian rhythmicity provide new avenues for the understanding and improvement of the sleep- wake cycle.
© Cambridge University Press 2013.In humans, the sleep–wake cycle is determined by the interaction of the endogenous circadian clock and sleep homeostat, and exogenous factors such as the light/dark cycle, which is important for circadian entrainment, and social influences such as work and recreation (Figure 31.1). These factors interact and it is often difficult to determine the causes and nature of altered sleep–wake timing. Abnormal sleep–wake timing may be a simple consequence of an abnormal phase relationship of the circadian clock and environmental time. This may be caused by aberrant light exposure patterns or extreme intrinsic periods of the circadian clock. The timing of the sleep–wake cycle relative to the circadian sleep propensity rhythm may be altered because of fast or slow build-up of homeostatic sleep pressure. Recent mathematical models of the sleep–wake cycle have indeed demonstrated that one particular phenotype may be related to parameters of very different processes [1]. Here, we focus on some of the genetic factors that are associated with abnormally delayed sleep timing, and explore to what extent the effects of these factors can be attributed to physiological processes such as light sensitivity, sleep homeostasis or circadian period. Circadian rhythm sleep disorders (CRSDs) refer to sleep disturbances that are primarily due to alterations of the circadian time-keeping system or are related to a misalignment of endogenous circadian rhythms and the required sleep–wake time (see [2]). The latter distinction is important because social factors may necessitate a non-desirable sleep–wake schedule, as occurs in shift work, for example. Shift work disorder and jet lag disorder are CRSDs that are caused by exogenous factors, whereas dysfunction of the endogenous circadian clock is thought to be the primary cause of delayed sleep phase disorder (DSPD). A better understanding of what causes CRSDs and inter-individual vulnerability differences is important because of the large proportion of the population who regularly undertake shift work, the epidemiological evidence linking insufficient sleep with negative health outcomes [3], and known associations between extreme evening preference and health problems such as mood disorders, metabolic disorders, and cardiovascular risk (see [4]).
Sleep and circadian rhythms are both considered to be important determinants of mental and physical health. Epidemiological studies characterise sleep by self-reported duration and quality whereas circadian aspects are often captured by self-reported chronotype. Several studies established the independent contribution of sleep duration, sleep quality or chronotype to health. A comparison of the relative contributions of sleep and circadian characteristics to health outcomes is currently not available. Mental health and sleep problems are more common in women than in men and men are more likely to be vening types than women. Few studies have explored sex differences in the associations between sleep-circadian characteristics and health. Here, sleep quality, chronotype, sleep duration and health and psychological wellbeing were assessed in 410 men and 261 women aged 18 to 30 by multiple questionnaires including the Morningness-Eveningness Questionnaire, Munich-ChronoType Questionnaire, Pittsburgh Sleep Quality Index, British Sleep Survey, Karolinska Sleep Diary, Insomnia Severity Index, SF-36 Health Survey, General Health Questionnaire, Dutch Eating-Behaviour Questionnaire, Big Five Inventory, Behaviour Inhibition System–Behaviour Activation System, and the Positive Affect-Negative Affect Schedule. Associations were assessed with multiple regression models and relative contributions of predictors were quantified as local effect sizes. Across all questionnaires, sleep quality was a much stronger independent predictor of health and in particular mental health than chronotype and sleep duration. The association between sleep quality and mental health was significant in both genders and significantly stronger in women than in men. A better understanding of subjective sleep quality may aid the understanding of sleep-health interactions.
Sleep disorders are common in the general population and even more so in clinical practice, yet are relatively poorly understood by doctors and other health care practitioners. These British Association for Psychopharmacology guidelines are designed to address this problem by providing an accessible up-to-date and evidence-based outline of the major issues, especially those relating to reliable diagnosis and appropriate treatment. A consensus meeting was held in London in May 2009. Those invited to attend included BAP members, representative clinicians with a strong interest in sleep disorders and recognized experts and advocates in the field, including a representative from mainland Europe and the USA. Presenters were asked to provide a review of the literature and identification of the standard of evidence in their area, with an emphasis on meta-analyses, systematic reviews and randomized controlled trials where available, plus updates on current clinical practice. Each presentation was followed by discussion, aimed to reach consensus where the evidence and/or clinical experience was considered adequate or otherwise to flag the area as a direction for future research. A draft of the proceedings was then circulated to all participants for comment. Key subsequent publications were added by the writer and speakers at draft stage. All comments were incorporated as far as possible in the final document, which represents the views of all participants although the authors take final responsibility for the document.
The circadian rhythms of melatonin and body temperature are set to an earlier hour in women than in men, even when the women and men maintain nearly identical and consistent bedtimes and wake times. Moreover, women tend to wake up earlier than men and exhibit a greater preference for morning activities than men. Although the neurobiological mechanism underlying this sex difference in circadian alignment is unknown, multiple studies in nonhuman animals have demonstrated a sex difference in circadian period that could account for such a difference in circadian alignment between women and men. Whether a sex difference in intrinsic circadian period in humans underlies the difference in circadian alignment between men and women is unknown. We analyzed precise estimates of intrinsic circadian period collected from 157 individuals (52 women, 105 men; aged 18-74 y) studied in a month-long inpatient protocol designed to minimize confounding influences on circadian period estimation. Overall, the average intrinsic period of the melatonin and temperature rhythms in this population was very close to 24 h [24.15 ± 0.2 h (24 h 9 min ± 12 min)]. We further found that the intrinsic circadian period was significantly shorter in women [24.09 ± 0.2 h (24 h 5 min ± 12 min)] than in men [24.19 ± 0.2 h (24 h 11 min ± 12 min); P < 0.01] and that a significantly greater proportion of women have intrinsic circadian periods shorter than 24.0 h (35% vs. 14%; P < 0.01). The shorter average intrinsic circadian period observed in women may have implications for understanding sex differences in habitual sleep duration and insomnia prevalence.
The high prevalence of sleep disturbance in neurodegenerative and psychiatric conditions is often interpreted as evidence for both sleep’s sensitivity to and causal involvement in brain pathology. Nevertheless, how and which aspects of sleep contribute to brain function remains largely unknown. This review provides a critical evaluation of clinical and animal literature describing sleep and circadian disturbances in two distinct conditions and animal models thereof: Alzheimer’s disease (AD) and schizophrenia. Its goal is to identify commonalities and distinctiveness of specific aspects of sleep disturbance and their relationship to symptoms across conditions. Despite limited standardisation, data imply that reductions in sleep continuity and alterations in sleep timing are common to AD and schizophrenia, whereas reductions in REM sleep and sleep spindle activity appear more specific to AD and schizophrenia, respectively. Putative mechanisms underlying these alterations are discussed. A standardised neuroscience based quantification of sleep and disease-independent assessment of symptoms in patients and animal models holds promise for furthering the understanding of mechanistic links between sleep and brain function in health and disease.
Human sleep schedules vary widely across countries. We investigated whether these variations were related to differences in social factors, Morningness-Eveningness (ME) preference, or the natural light-dark cycle by contrasting the sleep duration and timing of young adults (age: 18-35 years) on work and free days in Singapore (n = 1898) and the UK (n = 837). On work days, people in Singapore had later bedtimes, but wake times were similar to the UK sample, resulting in shorter sleep duration. In contrast, sleep duration on free days did not differ between the two countries. Shorter sleep on work days, without compensatory extra long sleep hours on free days, suggest greater demands from work and study in Singapore. While the two samples differed slightly in ME preference, the associations between eveningness preference and greater extension in sleep duration as well as delays in sleep timing on free days were similar in the two countries. Thus, differences in ME preference did not account for the differences in sleep schedules between the two countries. The greater variability in the photoperiod in the UK was not associated with more prominent seasonal changes in sleep patterns compared to Singapore. Furthermore, in the UK, daylight saving time did not alter sleep schedules relative to clock time. Collectively, these findings suggest that differences in social demands, primarily from work or study, could account for the observed differences in sleep schedules between countries, and that in industrialized societies, social zeitgebers, which typically involve exposure to artificial light, are major determinants of sleep schedules.
Slow waves (SWs, 0.5–4 Hz) in field potentials during sleep reflect synchronized alternations between bursts of action potentials and periods of membrane hyperpolarization of cortical neurons. SWs decline during sleep and this is thought to be related to a reduction of synaptic strength in cortical networks and to be central to sleep's role in maintaining brain function. A central assumption in current concepts of sleep function is that SWs during sleep, and associated recovery processes, are independent of circadian rhythmicity. We tested this hypothesis by quantifying all SWs from 12 EEG derivations in 34 participants in whom 231 sleep periods were scheduled across the circadian cycle in a 10-day forced-desynchrony protocol which allowed estimation of the separate circadian and sleep-dependent modulation of SWs. Circadian rhythmicity significantly modulated the incidence, amplitude, frequency and the slope of the SWs such that the peaks of the circadian rhythms in these slow-wave parameters were located during the biological day. Topographical analyses demonstrated that the sleep-dependent modulation of SW characteristics was most prominent in frontal brain areas whereas the circadian effect was similar to or greater than the sleep-dependent modulation over the central and posterior brain regions. The data demonstrate that circadian rhythmicity directly modulates characteristics of SWs thought to be related to synaptic plasticity and that this modulation depends on topography. These findings have implications for the understanding of local sleep regulation and conditions such as ageing, depression, and neurodegeneration which are associated with changes in SWs, neural plasticity and circadian rhythmicity.
OBJECTIVE: To explore the use of detrended fluctuation analysis (DFA) scaling exponent of the awake electroencephalogram (EEG) as a new alternative biomarker of neurobehavioural impairment and sleepiness in obstructive sleep apnea (OSA). METHODS: Eight patients with moderate-severe OSA and nine non-OSA controls underwent a 40-h extended wakefulness challenge with resting awake EEG, neurobehavioural performance (driving simulator and psychomotor vigilance task) and subjective sleepiness recorded every 2-h. The DFA scaling exponent and power spectra of the EEG were calculated at each time point and their correlation with sleepiness and performance were quantified. RESULTS: DFA scaling exponent and power spectra biomarkers significantly correlated with simultaneously tested performance and self-rated sleepiness across the testing period in OSA patients and controls. Baseline (8am) DFA scaling exponent but not power spectra were markers of impaired simulated driving after 24-h extended wakefulness in OSA (r=0.738, p=0.037). OSA patients had a higher scaling exponent and delta power during wakefulness than controls. CONCLUSIONS: The DFA scaling exponent of the awake EEG performed as well as conventional power spectra as a marker of impaired performance and sleepiness resulting from sleep loss. SIGNIFICANCE: DFA may potentially identify patients at risk of neurobehavioural impairment and assess treatment effectiveness.
Background: It has previously been reported that EEG sigma (10-15 Hz) activity during sleep exhibits infraslow oscillations (ISO) with a period of 50 seconds. However, a detailed analysis of the ISO of individually identified sleep spindles is not available. New Method: We investigated basic properties of ISO during baseline sleep of 34 healthy young human participants using a new and established methods. The analyses focused on fast sleep spindle and sigma activity (13-15 Hz) in NREM stage 2 and slow wave sleep (SWS). To describe ISO in sigma activity we analyzed power of power of the EEG signal. For the study of ISO in sleep spindle activity we applied a new method in which the EEG signal was reduced to a spindle on/off binary square signal. Its spectral properties were contrasted to that of a square signal wherein the same spindles and also the inter spindle intervals were permutated randomly. This approach was validated using surrogate data with imposed ISO modulation. Results: We confirm the existence of ISO in sigma activity albeit with a frequency below the previously reported 0.02 Hz. These ISO are most prominent in the high sigma band and over the centro-parieto-occipital regions. A similar modulation is present in spindle activity. ISO in sleep spindles are most prominent in the centro-parieto-occipital regions, left hemisphere and second half of the night independent of the number of spindles. Conclusions: The comparison of spectral properties of binary event signals and permutated event signals is effective in detecting slow oscillatory phenomena.
The phase and amplitude of rhythms in physiology and behavior are generated by circadian oscillators and entrained to the 24-h day by exposure to the light-dark cycle and feedback from the sleep-wake cycle. The extent to which the phase and amplitude of multiple rhythms are similarly affected during altered timing of light exposure and the sleep-wake cycle has not been fully characterized.
Slow wave sleep (SWS) has been reported to correlate with sleep maintenance, but whether pharmacological enhancement of SWS also leads to improved sleep maintenance is not known. Here we evaluate the time-course of the effects of gaboxadol, an extra-synaptic gamma-aminobutyric acid (GABA) agonist, on SWS, sleep maintenance, and other sleep measures in a traffic noise model of transient insomnia. After a placebo run-in, 101 healthy subjects (20-78 y) were randomized to gaboxadol (n = 50; 15 mg in subjects
Acute and chronic insufficient sleep are associated with adverse health outcomes and risk of accidents. There is therefore a need for biomarkers to monitor sleep debt status. None are currently available. We applied Elastic-net and Ridge regression to entire and pre-filtered transcriptome samples collected in healthy young adults during acute total sleep deprivation and following 1 week of either chronic insufficient (< 6 h) or sufficient sleep (~8.6 h) to identify panels of mRNA biomarkers of sleep debt status. The size of identified panels ranged from 9-74 biomarkers. Panel performance, assessed by leave-one-subject-out cross-validation and independent validation, varied between sleep debt conditions. Using between-subject assessments based on one blood sample, the accuracy of classifying ‘Acute sleep loss’ was 92%, but only 57% for classifying ‘Chronic sleep insufficiency’. A reasonable accuracy for classifying ‘chronic sleep insufficiency’ could only be achieved by a within-subject comparison of blood samples. Biomarkers for sleep debt status showed little overlap with previously identified biomarkers for circadian phase. Biomarkers for acute and chronic sleep loss also showed little overlap but were associated with common functions related to the cellular stress response, such as heat shock protein activity, the unfolded protein response, protein ubiquitination and endoplasmic reticulum associated protein degradation, and apoptosis. This characteristic response of whole blood to sleep loss can further aid our understanding of how sleep insufficiencies negatively affect health. Further development of these novel biomarkers for research and clinical practice requires validation in other protocols and age groups.
Reciprocal interactions between wakefulness and sleep substantially influence human brain function in both states of vigilance. On the one hand, there is evidence that regionally-specialized brain activity during wakefulness is modulated by the interaction between a local use-dependent buildup of homeostatic sleep pressure and circadian signals. On the other hand, brain activity during sleep, although mainly constrained by genuine sleep oscillations, shows wake-dependent regionally-specific modulations, which are involved in the dissipation of local homeostatic sleep pressure and memory consolidation.
Orexins have a role in sleep regulation, and orexin receptor antagonists are under development for the treatment of insomnia. We conducted a randomised, double-blind, placebo-controlled, four-period crossover study to investigate the effect of single doses of the dual orexin receptor antagonist SB-649868 (10 or 30 mg) and a positive control zolpidem (10 mg), an allosteric modulator of GABA(A) receptors. Objective and subjective sleep parameters and next-day performance were assessed in 51 healthy male volunteers in a traffic noise model of situational insomnia. Compared with placebo, SB-649868 10 and 30 mg increased total sleep time (TST) by 17 and 31 min (p
Light is a powerful modulator of cognition through its long-term effects on circadian rhythmicity and direct effects on brain function as identified by neuroimaging. How the direct impact of light on brain function varies with wavelength of light, circadian phase, and sleep homeostasis, and how this differs between individuals, is a largely unexplored area. Using functional MRI, we compared the effects of 1 minute of low-intensity blue (473 nm) and green light (527 nm) exposures on brain responses to an auditory working memory task while varying circadian phase and status of the sleep homeostat. Data were collected in 27 subjects genotyped for the PER3 VNTR (12 PER3(5/5) and 15 PER3(4/4) ) in whom it was previously shown that the brain responses to this task, when conducted in darkness, depend on circadian phase, sleep homeostasis, and genotype. In the morning after sleep, blue light, relative to green light, increased brain responses primarily in the ventrolateral and dorsolateral prefrontal cortex and in the intraparietal sulcus, but only in PER3(4/4) individuals. By contrast, in the morning after sleep loss, blue light increased brain responses in a left thalamofrontoparietal circuit to a larger extent than green light, and only so in PER3(5/5) individuals. In the evening wake maintenance zone following a normal waking day, no differential effect of 1 minute of blue versus green light was observed in either genotype. Comparison of the current results with the findings observed in darkness indicates that light acts as an activating agent particularly under those circumstances in which and in those individuals in whom brain function is jeopardized by an adverse circadian phase and high homeostatic sleep pressure.
Circadian rhythmicity and sleep homeostasis contribute to sleep phenotypes and sleep-wake disorders, some of the genetic determinants of which are emerging. Approximately 10% of the population are homozygous for the 5-repeat allele (PER3(5/5)) of a variable number tandem repeat polymorphism in the clock gene PERIOD3 (PER3). We review recent data on the effects of this polymorphism on sleep-wake regulation. PER3(5/5) are more likely to show morning preference, whereas homozygosity for the four-repeat allele (PER3(4/4)) associates with evening preferences. The association between sleep timing and the circadian rhythms of melatonin and PER3 RNA in leukocytes is stronger in PER3(5/5) than in PER3(4/4). EEG alpha activity in REM sleep, theta/alpha activity during wakefulness and slow wave activity in NREM sleep are elevated in PER3(5/5). PER3(5/5) show a greater cognitive decline, and a greater reduction in fMRI-assessed brain responses to an executive task, in response to total sleep deprivation. These effects are most pronounced during the late circadian night/early morning hours, i.e., approximately 0-4 h after the crest of the melatonin rhythm. We interpret the effects of the PER3 polymorphism within the context of a conceptual model in which higher homeostatic sleep pressure in PER3(5/5) through feedback onto the circadian pacemaker modulates the amplitude of diurnal variation in performance. These findings highlight the interrelatedness of circadian rhythmicity and sleep homeostasis. (C) 2009 Elsevier Ltd. All rights reserved.
In humans, sleep and wakefulness and the associated cognitive processes are regulated through interactions between sleep homeostasis and the circadian system. Chronic disruption of sleep and circadian rhythmicity is common in our society and there is a need for a better understanding of the brain mechanisms regulating sleep, wakefulness and associated cognitive processes. This review summarizes recent investigations which provide first neural correlates of the combined influence of sleep homeostasis and circadian rhythmicity on cognitive brain activity. Markers of interindividual variations in sleep-wake regulation, such as chronotype and polymorphisms in sleep and clock genes, are associated with changes in cognitive brain responses in subcortical and cortical areas in response to manipulations of the sleep-wake cycle. This review also includes recent data showing that cognitive brain activity is regulated by light, which is a powerful modulator of cognition and alertness and also directly impacts sleep and circadian rhythmicity. The effect of light varied with age, psychiatric status, PERIOD3 genotype and changes in sleep homeostasis and circadian phase. These data provide new insights into the contribution of demographic characteristics, the sleep-wake cycle, circadian rhythmicity and light to brain functioning.
Rationale Histamine and dopamine contribute to the maintenance of wakefulness. Objective This study aims to conduct an exploratory analysis of the effects of 10 and 50 mg of MK-0249, a novel histamine subtype-3 receptor inverse agonist, and 200 mg of modafinil, a presumed dopaminergic compound, on EEG power spectra during sleep deprivation and subsequent recovery sleep. Methods A total of 25 healthy men were recruited to a double-blind, placebo-controlled cross-over design. EEG power spectra, an electrophysiological marker of changes in sleepiness and vigilance, were obtained at the beginning of wake maintenance tests at two-hourly intervals throughout a night and day of sleep deprivation, which is an established model of excessive sleepiness. Results After placebo, sleep deprivation was associated with enhancements in delta and theta and reductions in alpha and beta activity. Following dosing at 02:00 h, MK-0249 and modafinil reduced delta and theta activity and enhanced alpha and beta activity, compared to placebo. During recovery sleep initiated at 21:00 h, latency to sleep onset and number of awakenings were not different from placebo for any of the active treatments. Wake after sleep onset and stage 1% was increased and total sleep time, SWS% and REM% were reduced after both doses of MK-0249. Compared to placebo, MK-0249, the 50-mg dose in particular, reduced activity in some delta and theta/alpha frequencies and enhanced beta activity during NREM sleep and REM sleep. After modafinil, no changes were observed for power spectra during sleep. Conclusion Both MK-0249 and modafinil exert effects on the EEG which are consistent with wake promotion.
Temporal organisation of molecular and physiological processes is driven by environmental and behavioural cycles, as well as by self-sustained molecular circadian oscillators. Quantification of phase, amplitude, period, and disruption of circadian rhythms is essential for understanding their contribution to sleep-wake disorders, social jet-lag, inter-individual differences in entrainment and the development of chrono-therapeutics. Traditionally, assessment of the human circadian system, and the output of the SCN in particular, required collection of long time series of univariate markers such as melatonin or core body temperature. Data were collected in specialised laboratory protocols designed to control for environmental and behavioural influences on rhythmicity. These protocols are time-consuming, expensive, and are not practical for assessing circadian status in patients or in participants in epidemiologic studies. Novel approaches for assessment of circadian parameters of the SCN or peripheral oscillators have been developed. They are based on machine learning or mathematical model-informed analyses of features extracted from one or a few samples of high dimensional data such as transcriptomes, metabolomes, long term simultaneous recording of activity, light exposure, skin temperature, and heart rate, or in vitro approaches. Here, we review whether these approaches successfully quantify parameters of central and peripheral circadian oscillators as indexed by gold standard markers. While several approaches perform well under entrained conditions when sleep occurs at night, the methods either perform worse in other conditions such as shift work, or they have not been assessed under any conditions other than entrainment and thus we do not yet know how robust they are. Novel approaches for the assessment of circadian parameters hold promise for circadian medicine, chrono-therapeutics, and chrono-epidemiology. There remains a need to validate these approaches against gold standard markers, in individuals of all sexes and ages, in patient populations, and, in particular, under conditions in which behavioural cycles are displaced.
Most antidepressants suppress rapid eye movement (REM) sleep, which is thought to be important to brain function, yet the resulting REM sleep restriction is well tolerated. This study investigated the impact of antidepressants with different mechanisms of action, such as selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants (TCA), on the regulation of REM sleep in rats. REM sleep was first demonstrated to be homeostatically regulated using 5, 8 and 10 hours of REM-sleep specific restriction through EEG-driven triggered arousals, with an average of 91 ± 10% of lost REM sleep recovered following a 26-29 -hour recovery period. Acute treatment with the antidepressants paroxetine, citalopram and imipramine inhibited REM sleep by 84 ± 8, 84 ± 8 and 69 ± 9% respectively relative to vehicle control. The pharmacologically-induced REM sleep deficits by citalopram and paroxetine were not fully recovered, whereas, after imipramine the REM sleep deficit was fully compensated. Given the marked difference between REM sleep recovery following the administration of paroxetine, citalopram, imipramine and REM sleep restriction, the homeostatic response was further examined by pairing REM sleep specific restriction with the three antidepressants. Surprisingly, the physiologically-induced REM sleep deficits incurred prior to suppression of REM sleep by all antidepressants was consistently recovered. The data indicate that REM sleep homeostasis remains operative following subsequent treatment with antidepressants and is unaffected by additional pharmacological inhibition of REM sleep.
One of sleep’s putative functions is mediation of adaptation to waking experiences. Chronic stress is a common waking experience, however, which specific aspect of sleep is most responsive, and how sleep changes relate to behavioral disturbances and molecular correlates remain unknown. We quantified sleep, physical, endocrine and behavioral variables, as well as the brain and blood transcriptome in mice exposed to nine weeks of unpredictable chronic mild stress (UCMS). Comparing 46 phenotypical variables revealed that rapid-eye-movement sleep (REMS), corticosterone regulation and coat state were most responsive to UCMS. REMS theta oscillations were enhanced whereas delta oscillations in non-REMS were unaffected. Transcripts affected by UCMS in the prefrontal cortex, hippocampus, hypothalamus and blood were associated with inflammatory and immune responses. A machine learning approach controlling for unspecific UCMS effects identified transcriptomic predictor sets for REMS parameters which were enriched in 193 pathways, including some involved in stem cells, immune response, apoptosis and survival. Only 3 pathways were enriched in predictor sets for non-REMS. Transcriptomic predictor sets for variation in REMS continuity and theta activity shared many pathways with corticosterone regulation, in particular pathways implicated in apoptosis and survival, including mitochondrial apoptotic machinery. Predictor sets for REMS and anhedonia shared pathways involved in oxidative stress, cell proliferation and apoptosis. These data identify REMS as a core and early element of the response to chronic stress, and identify apoptosis and survival pathways as a putative mechanism by which REMS may mediate the response to stressful waking experiences.
Sleep is regulated by a homeostatic process which in the two-process model of human sleep regulation is represented by EEG slow-wave activity (SWA). Many studies of acute manipulation of wake duration have confirmed the precise homeostatic regulation of SWA in rodents and humans. However, some chronic sleep restriction studies in rodents show that the sleep homeostatic response, as indexed by SWA, is absent or diminishes suggesting adaptation occurs. Here, we investigate the response to 7 days of sleep restriction (6 h time in bed) and extension (10 h time in bed) as well as the response to subsequent total sleep deprivation in 35 healthy participants in a cross-over design. The homeostatic response was quantified by analyzing sleep structure and SWA measures. Sleep restriction resulted primarily in a reduction of REM sleep. SWA and accumulated SWA (slow-wave energy) were not much affected by sleep extension/restriction. The SWA responses did not diminish significantly in the course of the intervention and did not deviate significantly from the predictions of the two-process model. The response to total sleep deprivation consisted of an increase in SWA, rise rate of SWA and SWE and did not differ between the two conditions. The data show that changes in sleep duration within an ecologically relevant range, have a marked effect on REM sleep and that SWA responds in accordance with predictions based on a saturating exponential increase during wake and an exponential decline in sleep of homeostatic sleep pressure during both chronic sleep restriction and extension.
Circadian dysregulation causes sleep disturbance and impacts quality of life and functioning. Some interventions target circadian entrainment through modifying light exposure, but existing reviews of light interventions for sleep improvement include few studies in psychiatric populations. We examined effect of light interventions on sleep quality, duration and timing, and effect moderators. We included controlled studies in intrinsic circadian rhythm disorders (such as advanced or delayed sleep) and in neuropsychiatric disorders with assumed high prevalence of circadian dysregulation (such as affective and psychotic disorders). Articles were identified through database searching: 40 studies reporting 49 relevant intervention comparisons met inclusion criteria. Meta-analysis showed improvements in sleep continuity (ES = −0.23, p = 0.000), self-reported sleep disturbance (ES = −0.32, p = 0.014), and advancement of delayed sleep timing (ES = −0.34, p = 0.010). Although the small number of studies limited meta-regression, evening light avoidance was associated with greater increase in total sleep time. Effects of light on sleep and circadian outcomes have received limited attention in studies in psychiatric disorders, but results were promising in these groups. These findings invite further refinement and testing of light interventions to improve sleep in psychiatric disorders, with improved assessment and specification of problems, and the development and implementation of light schedule interventions for delayed sleep.
Light is considered the most potent synchronizer of the human circadian system and exerts many other non-image-forming effects, including those that affect brain function. These effects are mediated in part by intrinsically photosensitive retinal ganglion cells that express the photopigment melanopsin. The spectral sensitivity of melanopsin is greatest for blue light at approximately 480 nm. At present, there is little information on how the spectral composition of light to which people are exposed varies over the 24 h period and across seasons. Twenty-two subjects, aged 22±4 yrs (mean±SD) participated during the winter months (November–February), and 12 subjects aged 25±3 yrs participated during the summer months (April–August). Subjects wore Actiwatch-RGB monitors, as well as Actiwatch-L monitors, for seven consecutive days while living in England. These monitors measured activity and light exposure in the red, green, and blue spectral regions, in addition to broad-spectrum white light, with a 2 min resolution. Light exposure during the day was analyzed for the interval between 09:00 and 21:00 h. The time course of white-light exposure differed significantly between seasons (p = 0.0022), with light exposure increasing in the morning hours and declining in the afternoon hours, and with a more prominent decline in the winter. Overall light exposure was significantly higher in summer than winter (p = 0.0002). Seasonal differences in the relative contribution of blue-light exposure to overall light exposure were also observed (p = 0.0006), in particular during the evening hours. During the summer evenings (17:00–21:00 h), the relative contribution of blue light was significantly higher (p
Cognition is regulated across the 24 h sleep-wake cycle by circadian rhythmicity and sleep homeostasis through unknown brain mechanisms. We investigated these mechanisms in a functional magnetic resonance imaging study of executive function using a working memory 3-back task during a normal sleep-wake cycle and during sleep loss. The study population was stratified according to homozygosity for a variable-number (4 or 5) tandem-repeat polymorphism in the coding region of the clock gene PERIOD3. This polymorphism confers vulnerability to sleep loss and circadian misalignment through its effects on sleep homeostasis. In the less-vulnerable genotype, no changes were observed in brain responses during the normal-sleep wake cycle. During sleep loss, these individuals recruited supplemental anterior frontal, temporal and subcortical regions, while executive function was maintained. In contrast, in the vulnerable genotype, activation in a posterior prefrontal area was already reduced when comparing the evening to the morning during a normal sleep-wake cycle. Furthermore, in the morning after a night of sleep loss, widespread reductions in activation in prefrontal, temporal, parietal and occipital areas were observed in this genotype. These differences occurred in the absence of genotype-dependent differences in circadian phase. The data show that dynamic changes in brain responses to an executive task evolve across the sleep-wake and circadian cycles in a regionally specific manner that is determined by a polymorphism which affects sleep homeostasis. The findings support a model of individual differences in executive control, in which the allocation of prefrontal resources is constrained by sleep pressure and circadian phase.
Study Objectives. Sleep restriction leads to performance decrements across cognitive domains but underlying mechanisms remain largely unknown. The impact of sleep restriction on performance in rodents is often assessed using tasks in which food is the reward. Investigating how the drives of hunger and sleep interact to modulate performance may provide insights into mechanisms underlying sleep loss related performance decrements. Methods. Three experiments were conducted in male adult Wistar rats to assess: 1) Effects of food restriction on performance in the simple response latency task (SRLT) across the diurnal cycle (n=30); 2) Interaction of food restriction and sleep restriction (11-h) on SRLT performance, sleep EEG and event-related potentials (ERP) (n=10-13); 3) Effects of food restriction and sleep restriction on progressive ratio (PR) task performance to probe the reward value of food reinforcement (n=19). Results. Food restriction increased premature responding on the SRLT at the end of the light period of the diurnal cycle. Sleep restriction led to marked impairments in SRLT performance in the ad libitum-fed group, which were absent in the food-restricted group. After sleep restriction, food restricted rats displayed a higher amplitude of cue-evoked ERP components during the SRLT compared to the ad libitum group. Sleep restriction did not affect PR performance while food restriction improved performance. Conclusions. Hunger may induce a functional resilience to negative effects of sleep loss during subsequent task performance, possibly by maintaining attention to food-related cues.
In humans, slow-wave sleep (SWS) consists of stages 3 and 4 of non rapid eye movement (nonREM) sleep. The low-frequency, high-amplitude slow waves that dominate the electroencephalogram (EEG) during SWS can be quantified as slow-wave activity (SWA). SWS and SWA are regulated very accurately in response to variations in the duration and intensity of wakefulness and sleep. SWA declines more or less independently of circadian phase during the course of a sleep episode, indicating that it is primarily under homeostatic rather than circadian control. An age-related decline in SWS and SWA is well established. In some studies, apprehension, depression and insomnia have been associated with reductions in SWS and SWA. Experimental reductions of SWS through SWS deprivation (without altering total sleep time or REM duration) have been reported to lead to an increase in daytime sleep propensity and reductions in performance. SWS and SWA are therefore thought to contribute to the recovery processes that occur during sleep. Most currently prescribed hypnotics, such as the benzodiazepines and Z-drugs, suppress SWA. Some compounds have been shown to enhance SWS and SWA in healthy volunteers through GAT-1 inhibition, GABA-A modulation, GABA-B modulation, and 5HT2(A) antagonism. Pharmacological enhancement of SWS has also been observed in insomnia. The effects of SWS enhancement on other sleep parameters will be discussed.
Since the discovery of the antidepressant effects of interventions in the sleep-wake cycle, a number of hypotheses have emerged according to which disturbances in sleep physiology are not merely expressions but essential components of the pathophysiology of depression. Three hypotheses are presented, the "Phase-advance", the "S-deficiency" and the "ACh-hypersensitivity" hypotheses. They explain the therapeutic effects of total, partial and selective sleep deprivation as consequences of the normalization of disturbed sleep regulation. The question is dealt with whether there are indications that the hypothesized sleep regulatory disturbances exist and whether there is a relationship between the effects of sleep deprivation on sleep regulation and clinical state. None of the hypotheses is totally supported, none can be fully rejected.
In organisms as diverse as single-celled algae and humans, light is the primary stimulus mediating entrainment of the circadian biological clock. Reports that some totally blind individuals appear entrained to the 24-h day have suggested that nonphotic stimuli may also be effective circadian synchronizers in humans, although the nonphotic stimuli are probably comparatively weak synchronizers, because the circadian rhythms of many totally blind individuals "free run" even when they maintain a 24-h activity-rest schedule. To investigate entrainment by nonphotic synchronizers, we studied the endogenous circadian melatonin and core body temperature rhythms of 15 totally blind subjects who lacked conscious light perception and exhibited no suppression of plasma melatonin in response to ocular bright-light exposure. Nine of these fifteen blind individuals were able to maintain synchronization to the 24-h day, albeit often at an atypical phase angle of entrainment. Nonphotic stimuli also synchronized the endogenous circadian rhythms of a totally blind individual to a non-24-h schedule while living in constant near darkness. We conclude that nonphotic stimuli can entrain the human circadian pacemaker in some individuals lacking ocular circadian photoreception.
The effect of sleep deprivation (40 h) on topographic and temporal aspects of electroencephalographic (EEG) activity during sleep was investigated by all night spectral analysis in six young volunteers. The sleep-deprivation-induced increase of EEG power density in the delta and theta frequencies (1-7 Hz) during nonREM sleep, assessed along the antero-posterior axis (midline: Fz, Cz, Pz, Oz), was significantly larger in the more frontal derivations (Fz, Cz) than in the more parietal derivations (Pz, Oz). This frequency-specific frontal predominance was already present in the first 30 min of recovery sleep, and dissipated in the course of the 8-h sleep episode. The data demonstrate that the enhancement of slow wave EEG activity during sleep following extended wakefulness is most pronounced in frontal cortical areas.
We argue that by neglecting the fact that procedural memory may also have episodic qualities, and by considering only a systems approach to memory, Walker's account of consolidation of learning during subsequent sleep ignores alternative accounts of how sleep stages may be interdependent. We also question the proposition that sleep-based consolidation largely bypasses hippocampal structures.
The contribution of the circadian timing system to the age-related advance of sleep-wake timing was investigated in two experiments. In a constant routine protocol, we found that the average wake time and endogenous circadian phase of 44 older subjects were earlier than that of 101 young men. However, the earlier circadian phase of the older subjects actually occurred later relative to their habitual wake time than it did in young men. These results indicate that an age-related advance of circadian phase cannot fully account for the high prevalence of early morning awakening in healthy older people. In a second study, 13 older subjects and 10 young men were scheduled to a 28-h day, such that they were scheduled to sleep at many circadian phases. Self-reported awakening from scheduled sleep episodes and cognitive throughput during the second half of the wake episode varied markedly as a function of circadian phase in both groups. The rising phase of both rhythms was advanced in the older subjects, suggesting an age-related change in the circadian regulation of sleep-wake propensity. We hypothesize that under entrained conditions, these age-related changes in the relationship between circadian phase and wake time are likely associated with self-selected light exposure at an earlier circadian phase. This earlier exposure to light could account for the earlier clock hour to which the endogenous circadian pacemaker is entrained in older people and thereby further increase their propensity to awaken at an even earlier time.
Study Objectives: Individual sleep timing differs and is governed partly by circadian oscillators, which may be assessed by hormonal markers, or by clock gene expression. Clock gene expression oscillates in peripheral tissues, including leukocytes. The study objective was to determine whether the endogenous phase of these rhythms, assessed in the absence of the sleep-wake and light-dark cycle, correlates with habitual sleep-wake timing. Design: Observational, cross-sectional. Setting: Home environment and Clinical Research Center. Participants: 24 healthy subjects aged 25.0 ± 3.5 (SD) years. Measurements: Actigraphy and sleep diaries were used to characterize sleep timing. Circadian rhythm phase and amplitude of plasma melatonin, cortisol, and BMAL1, PER2, and PER3 expression were assessed during a constant routine. Results: Circadian oscillations were more robust for PER3 than for BMAL1 or PER2. Average peak timings were 6:05 for PER3, 8:06 for PER2, 15:06 for BMAL1, 4:20 for melatonin, and 10:49 for cortisol. Individual sleep-wake timing correlated with the phases of melatonin and cortisol. Individual PER3 rhythms correlated significantly with sleep-wake timing and the timing of melatonin and cortisol, but those of PER2 and BMAL1 did not reach significance. The correlation between sleep timing and PER3 expression was stronger in individuals homozygous for the variant of the PER3 polymorphism that is associated with morningness. Conclusions: Individual phase differences in PER3 expression during a constant routine correlate with sleep timing during entrainment. PER3 expression in leukocytes represents a useful molecular marker of the circadian processes governing sleep-wake timing.
In eight healthy middle-aged men, sleep and core body temperature were recorded under baseline conditions, during all-night SWS suppression by acoustic stimulation, and during undisturbed recovery sleep. SWS suppression resulted in a marked reduction of sleep stages 3 and 4 but did not affect the time course of core body temperature. These data suggest that sleep stages 3 and 4 of nonREM sleep (i.e. SWS) do not play a major role in the regulation of core body temperature in humans.
The relation between the duration of prior wakefulness and EEG power density during sleep in humans was assessed by means of a study of naps. The duration of prior wakefulness was varied from 2 to 20 hr by scheduling naps at 1000 hr, 1200 hr, 1400 hr, 1600 hr, 1800 hr, 2000 hr, and 0400 hr. In contrast to sleep latencies, which exhibited a minimum in the afternoon, EEG power densities in the delta and theta frequencies were a monotonic function of the duration of prior wakefulness. The data support the hypothesis that EEG power density during non-rapid eye movement sleep is only determined by the prior history of sleep and wakefulness and is not determined by clock-like mechanisms.
The sensitivity of the human circadian system to light has been the subject of considerable debate. Using computer simulations of a recent quantitative model for the effects of light on the human circadian system, we investigated these effects of light during different experimental protocols. The results of the simulations indicate that the nonuniform distribution over the circadian cycle of exposure to ordinary room light seen in classical free-run studies, in which subjects select their exposure to light and darkness, can result in an observed period of approximately 25 h, even when the intrinsic period of the subject's endogenous circadian pacemaker is much closer to 24 h. Other simulation results suggest that accurate assessment of the true intrinsic period of the human circadian pacemaker requires low ambient light intensities (approximately 10-15 lx) during scheduled wake episodes, desynchrony of the imposed light-dark cycle from the endogenous circadian oscillator, and a study length of at least 20 days. Although these simulations await further experimental substantiation, they highlight the sensitivity to light of the human circadian system and the potential confounding influence of light on the assessment of the intrinsic period of the circadian pacemaker.
Increasing sleep pressure is associated with highly predictable changes in the dynamics of the sleep electroencephalogram (EEG). To investigate whether the effects of reduced sleep pressure also can be accounted for by homeostatic mechanisms, nighttime sleep following an evening nap was recorded in healthy young men. In comparison with the baseline night, sleep latency in the postnap night was prolonged, rapid eye movement sleep (REMS) latency was reduced, and EEG power density in non-REMS was decreased in the delta and theta band. The buildup of both EEG slow-wave activity (SWA; power density in the 0.75-to 4.5-Hz range) and spindle frequency activity (SFA; power density in the 12.25-to 15.0-Hz range) in non-REMS episodes was diminished (SWA: episodes 1-3; SFA: episode 1). The typical declining trend of SWA over consecutive non-REM sleep episodes was attenuated. The time course of SWA could be closely simulated with a homeostatic model of sleep regulation, although some discrepancies in level and buildup of SWA were apparent. We conclude that homeostatic mechanisms can largely account for the dynamics of the sleep EEG under conditions of reduced sleep pressure.
Circadian and sleep/wake dependent processes underlying variations in subjective alertness and cognitive performance were assessed in a constant routine protocol and in a protocol in which the sleep/wake cycle was uncoupled from the output of the endogenous circadian pacemaker. In the latter protocol, the contribution of a sleep/wake dependent process and a circadian process to alertness and performance were separated by folding the data at either the period of the sleep/wake cycle or at the period of the endogenous circadian body temperature rhythm. This analysis revealed that prior wakefulness within a range of 0-18 h significantly reduced alertness and performance and that the circadian rhythm of core body temperature paralleled the circadian rhythm of alertness and performance. During the first 16 h of the constant routine protocol, which coincided with the subjects' habitual period of wakefulness, alertness and performance remained at a stable level. The latter finding was explained by assuming that during our usual waking day the circadian system counteracts the detrimental effects of increasing duration of prior wakefulness.