Professor Joseph Keddie FInstP
About
Biography
Joseph (Joe) Keddie is Professor of Soft Matter Physics within the Soft Matter, Biological and Medical Physics Group at the University of Surrey. He is also a Royal Society Industry Fellow and a Fellow of the Institute for Sustainability at Surrey. He leads a vibrant research programme in soft matter physics.
After graduating with a PhD from Cornell University (USA) in 1993, Joe spent two years as a post-doctoral researcher in the Polymer & Colloids Group at the Cavendish Laboratory, University of Cambridge followed by one year as an Oppenheimer Fellow there . At Cambridge, he used spectroscopic ellipsometry and environmental-SEM to study polymer thin films and colloids. In 1995 Joe moved to the University of Surrey as a Foundation Fund lecturer. Throughout his time at Surrey, he has carried out research on soft matter, with special interest in polymer colloids. More recently, research has focussed on sustainable materials and living materials.
In recognition of his contributions to the understanding of polymer dynamics of polymers in thin films and colloidal dispersions, he was awarded the 2001 Paterson Medal and Prize by the Institute of Physics and named a Fellow. His research group has won two Roon Awards from the Federation of Societies for Coatings Technology for papers presented at their Annual Meeting. In 2006 he was inducted into the Phi Beta Kappa honour society by his alma mater, Alfred University. He was selected to deliver the 2017 Thomas Graham Lecture by the RSC/SCI Joint Colloids Group. He held a Royal Society Short Industry Fellowship in 2023 and commenced a four-year Royal Society Industry Fellowship in 2024.
A co-author of >150 journal articles and book chapters, he also holds two US patents and has recently submitted two international patent applications. He has written - along with his co-author, Alex Routh, at the University of Cambridge - a book entitled Fundamentals of Latex Film Formation: Processes and Properties, which was published in 2010 by Springer. His current h-index is 53 (according to Google Scholar).
From 2011 to 2017, he served as the chair of the Polymer Physics Group of the Institute of Physics. He was recently the Awards Co-ordinator for the RSC/SCI Joint Colloids Group.
Areas of specialism
University roles and responsibilities
- Senator representing the Faculty of Engineering and Physical Sciences
- Senior Tutor for Professional Training for Physics
My qualifications
BA in English
Affiliations and memberships
Business, industry and community links
News
In the media
ResearchResearch interests
My research team explores relationships between the structure and properties of soft matter systems. We also investigate fundamental processes in polymer colloids, including drying, ordering, coalescence, and water transport.
We make use of a numerous non-invasive techniques to characterise soft matter. We frequently employ NMR profiling, atomic force microscopy, UV-Vis-NIR ellipsometry (with a brand-new instrument), and thermomechanical analysis. We have recently used national facilities for small-angle neutron scattering and ion beam analysis (Rutherford backscattering and elastic recoil detection). Recently, we have been investigating the suitability of super-resolution optical microscopy to soft matter systems.
We study a variety of variety of soft matter systems. Recent examples are:
- Biocoatings: Bacteria confined in colloidal coatings
- Pickering emulsions stabilised with nanodiamonds, peptides, and hybrids.
- Polymorphs of amino acid crystals
- Physically and chemically crosslinked gelatin
- 2-D materials (MoS2) in polymers
- Carbon nanotubes and graphene oxide in polymers
- Surfactant-free polymer colloids
We develop soft matter for applications. Recent examples are:
- Biocoatings for carbon capture, oxygen production, and wastewater treatment
- Soft adhesives: debond-on-demand and degradable
- Switchable pressure-sensitive adhesives
- Self-stratifying coatings
- Plasmonic and photonic materials made using Au nanoparticles
- Fibronectin-nanopatterned substrates for cell growth
- Radiation detectors from semiconducting polymers and small organic molecules
Research projects
Biocoatings contain nongrowing, metabolically-active bacteria in a polymer binder where they perform useful functions, such as absorbing greenhouse gases (CO2, methane) and producing biomass or gases for fuel cells (H2, O2). We will solve problems with biocoatings that limit their effectiveness. To enable transport of gases and products within the coatings, we will introduce natural clay nanotubes (halloysite) to create nanoporous channels. To impart uniform hydrophilicity and to reduce drying stresses on the bacteria, we will employ natural nanoparticles based on glucose (phytoglycogen) in the structures. To increase adhesion of the cells to the coating, we will introduce charge attractions.
Funded by The Leverhulme Trust (42 months from April 2019)
Self-Stratification of Bimodal Linear and Star Polymers during the Drying of Thin FilmsPolymers are large molecules, typically synthesized from petroleum sources. The size of the polymer molecules has an effect on the adhesion to the surface and the temperature at which it softens. This project aims to answer the simple question: if a mixture of large and small linear polymers of the same composition are deposited from a solution to make a film, will they be randomly mixed, or will one of the chain populations be enriched at the surface? Polymers can also be synthesized in the shape of a star, with arms extending from a central core. We will also aim to answer the question: if a mixture of large and small star molecules (or linear and star polymers) is cast from a solution, will the small molecules be enriched at the surface? This project will provide excellent training for two undergraduates working closely with a post-doctoral researcher. To answer these questions, a student will model the non-equilibrium systems. In experiments, students will scatter high energy ions from the polymer films to determine their composition as a function of depth. The extent of layering or stratification will be precisely measured. The research will provide new understanding of ways to control surface properties of polymers.
Funded by the ACS Petroleum Research Fund (Sept. 2019 - August 2021)
Steering Colloids via Two-Dimensional Diffusiophoresis Using Crossed Gradients in Salt ConcentrationsColloidal particles in liquids are found in many ordinary items, such as foods, paints, inks, cosmetics, and pharmaceuticals. Colloids in liquid are not static, but they are constantly moving in random directions because of Brownian diffusion. In this research, we will investigate ways to move colloids in water in specific desired directions.
When colloids have an electric charge they can be steered by an external electric field acting upon them through the mechanism known as electrophoresis. Recently, scientists have discovered that the electric fields created by salts dissolved non-uniformly in water can drive colloid motion. The colloidal particles will move up or down a gradient in salt concentration, depending on the type of salt and whether the charge on the particles is positive or negative.
Our vision in this project is to steer particles on curved paths by putting them in solutions in which there are gradients of two different salts going in directions at right angles to each other. We will use criss-crossed polymeric fibres to release salts into water as a way to devise complex patterns of salt concentrations. According to some recent calculations, it should be possible in these salt gradients to separate mixtures of particles that differ in their charge. This new concept will allow particles to be sorted in by a simple, low-cost method. An immediate application will be in re-using and recycling expensive colloids to minimise waste. The use of crossed gradients in salt concentration could also provide a way to measure the unknown electric charge on particles. The cross gradients of salt concentrations will provide a simple diagnostic method to identify particles, such as viruses or contaminants.
Having such precise control of colloid motion will open up possibilities for fabricating complex materials one particle at a time. We will attach some “sticky” molecules onto the colloids so that they will attach to surfaces after being steered their by electrophoresis. Our fundamental research might thereby lead to breakthroughs in the manufacturing of materials for applications in photonics, electronic devices or drug delivery.
Funded by EPSRC New Horizons Programme (February 2021 - November 2022)
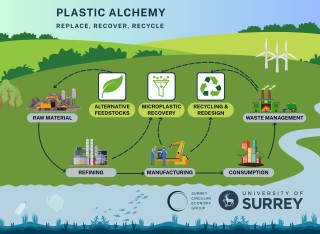
Next-generation sustainable liquid food packaging using only bio-based feedstock derived from non-food wasteThe ‘Pulpex UK Bioscience Initiative’ will help Pulpex Ltd fully achieve its medium- term ambition by replacing its existing petrochemical-based barrier coatings with truly sustainable coatings as barriers to water and foods. The barrier coatings are applied on the interior of the Pulpex sustainable bottle. Sustainable materials are the future of food packaging; consumers, general public and corporates are demanding this product now.
Pulpex has already demonstrated the manufacture of a paper bottle that will enable brands to switch from glass or plastic to a sustainable alternative that is readily recycled. Pulpex has developed an advanced manufacturing process that converts cellulose fibre into a bottle that can be easily recycled through the existing and well-established paper collection schemes that are available throughout the UK.
Pulpex are partnering with two UK biomanufacturing firms. CuanTec is a world leading firm based in the European Centre for Marine Biotechnology in Oban who produce chitosan from waste material from shellfish using a proprietary fermentation process. Cellucomp is an established company based in Fife with deep expertise in the extraction of micro- fibrillated cellulose (MFC) from waste root vegetables using bioprocessing techniques. These are world-class companies based here the UK and seeking expanding markets for their products.
This ambitious collaborative project draws on scientific expertise from across the UK to accelerate the adoption of bioprocessing-derived barrier coatings for Pulpex’s breakthrough fibre bottles and other high barrier packaging applications. The incredible calibre of our partners is testament to the high standards we aspire to. The project offers deep industry and academic collaboration across the entire supply chain – converting non-food waste (e.g. shells from crustaceans and discards from root vegetables) into high value high barrier packaging that is market ready and validated. We see properly bioprocessing as essential to the development of the next generation of sustainable products.
The project aim is to deliver and accelerate commercial high barrier products – derived from the bioprocessing / fermentation of chitosan and MFC from vegetal waste streams – that can be effectively applied to Pulpex fibre bottles at scale and certified as suitable for liquid food use.
Funded by Innovate UK Biomanufacturing (Sept. 2023 - Aug. 2025)
Improving the Barrier Properties of Coatings on Wood Pulp Bottles for the Reduction of Plastic WasteHumans currently produce approximately 583 billion plastic bottles every year, with 38.5 million being used daily in the UK. Of this number in the UK, 39% are not recycled and 2% are littered, with devastating impact on soil, water and wildlife. A new approach to solve the problem of waste plastic packaging is offered by bottles made from wood pulp. In a process developed by Pulpex, wood-pulp bottles are sprayed internally with specialised coatings designed to be a barrier to prevent liquids and gases from seeping through the walls.
Pulpex bottles are made from sustainably sourced, FSC-certified wood pulp. Bottles can be recycled in the same way as paper and card. In the event that the bottles are not recycled or correctly disposed of, they will degrade readily in the natural environment. The carbon footprint for wood pulp bottles is 30% less than for PET.
Coatings inside the bottles are deposited from a wet mixture of different types of particles. The barrier properties of the coatings are easily affected by small changes in the drying conditions. This research will test the hypothesis that when the evaporation of water across a surface is non-uniform, pH gradients and particle flow will be induced within the coating, ultimately leading to a degradation of properties.
Royal Society Short Industry Fellowship (Oct. 2022 - Aug. 2023)
Truly Sustainable Materials for the Packaging of LiquidsApproximately 38.5 million plastic bottles are used daily in the UK. Of this number, 39% are not recycled, and unfortunately 2% are littered, with devastating impact on soil, water and wildlife. Although individuals regret the environmental impact of plastics, and the high energy demand used in glass manufacturing, they have few alternatives when buying food and drink, cosmetics or homecare products.
To address this problem, Pulpex Ltd has demonstrated the manufacture of a paper bottle that will enable commercial brands to switch their packaging from glass or plastic to a sustainable alternative. Pulpex has developed an advanced manufacturing process that converts cellulose fibre into a bottle that can be easily recycled through existing, well-established paper collection schemes. Moreover, the carbon footprint for these paper bottles is 30% less than for PET. The Pulpex bottle presently uses a petrochemical-based coating to provide a physical barrier to contain liquids. Pulpex has a medium-term ambition to introduce truly sustainable, natural materials that will degrade harmlessly in the environment, if not recycled with paper.
Professor Keddie will apply his lifetime’s knowledge of coatings to develop sustainable barrier materials for the Pulpex bottle. He will focus on chitosan, a natural material derived from crustaceans. Chitosan will be modified so that liquids will permeate much more slowly through coatings made from it. Using physical chemistry principles, multiple layers will be engineered to make liquid flow less favourable. Ultimately, these coatings will be applied to Pulpex paper bottles at scale and certified for liquid food use.
Royal Society Industry Fellowship (Sept. 2024 - Aug. 2028)
Research collaborations
Our research is inter-disciplinary, and so we collaborate regularly with chemists, who synthesise molecules and particles, with microbiologists with expertise in bacteria, and with engineers with expertise in applications. Our work is appreciated and supported by industry.
- Previous EC-funded projects include
BarrierPlus: developed waterborne, surfactant-free barrier coatings for steel
NAPOLEON: produced waterborne polymer nanocomposites for applications in adhesives, coatings and cosmetics.
NSHAPE: designed core-shell, crosslinking polymer particles for pressure-sensitive adhesives
- Recent collaborations with resulting joint publications with
- Dr Cecile Dreiss (King's College London)
- Prof. Alex Routh (University of Cambridge)
- Prof. Jose Asua (POLYMAT, University of the Basque Country, UPV/EHU)
- Prof. Stefan Bon (University of Warwick)
- Prof. Alan Dalton (University of Sussex)
- Prof. Otto Muskens and Dr. Antonios Kanaras (University of Southampton)
- Prof. Elodie Bourgeat-Lami, Frank D'Agosto, and Muriel Lansalot (Univ. Lyon)
- Dr. Suzie Hingley-Wilson (University of Surrey)
- Current industrial collaborations with Avery Dennison and Syngenta.
- Previous industrial collaborations with Synthomer, BASF, DSM, Allnex, AkzoNobel, and International Paint.
Royal Society Industry Fellowship with a 50% secondment with Pulpex Ltd, developing sustainable packaging for liquids.
Indicators of esteem
2017 Thomas Graham Lecturer awarded by the RSC/SCI Joint Colloids Group
2015 Medallion from Coatings Societies International
Fellow of the Institute of Physics
International Scientific Advisory Committee of Advanced Research Center for Chemical Building Blocks in the Netherlands
Editorial Advisory Board of ACS Applied Materials and Interfaces
Awards Co-ordinator for the RSC/SCI Joint Colloids Group
Trustee of the High Polymer Research Group
2001 Winner of the Paterson Medal and Prize from the Institute of Physics for “major contributions to the understanding of the dynamics of polymers at surfaces, in thin films and in colloid dispersions”
Research interests
My research team explores relationships between the structure and properties of soft matter systems. We also investigate fundamental processes in polymer colloids, including drying, ordering, coalescence, and water transport.
We make use of a numerous non-invasive techniques to characterise soft matter. We frequently employ NMR profiling, atomic force microscopy, UV-Vis-NIR ellipsometry (with a brand-new instrument), and thermomechanical analysis. We have recently used national facilities for small-angle neutron scattering and ion beam analysis (Rutherford backscattering and elastic recoil detection). Recently, we have been investigating the suitability of super-resolution optical microscopy to soft matter systems.
We study a variety of variety of soft matter systems. Recent examples are:
- Biocoatings: Bacteria confined in colloidal coatings
- Pickering emulsions stabilised with nanodiamonds, peptides, and hybrids.
- Polymorphs of amino acid crystals
- Physically and chemically crosslinked gelatin
- 2-D materials (MoS2) in polymers
- Carbon nanotubes and graphene oxide in polymers
- Surfactant-free polymer colloids
We develop soft matter for applications. Recent examples are:
- Biocoatings for carbon capture, oxygen production, and wastewater treatment
- Soft adhesives: debond-on-demand and degradable
- Switchable pressure-sensitive adhesives
- Self-stratifying coatings
- Plasmonic and photonic materials made using Au nanoparticles
- Fibronectin-nanopatterned substrates for cell growth
- Radiation detectors from semiconducting polymers and small organic molecules
Research projects
Biocoatings contain nongrowing, metabolically-active bacteria in a polymer binder where they perform useful functions, such as absorbing greenhouse gases (CO2, methane) and producing biomass or gases for fuel cells (H2, O2). We will solve problems with biocoatings that limit their effectiveness. To enable transport of gases and products within the coatings, we will introduce natural clay nanotubes (halloysite) to create nanoporous channels. To impart uniform hydrophilicity and to reduce drying stresses on the bacteria, we will employ natural nanoparticles based on glucose (phytoglycogen) in the structures. To increase adhesion of the cells to the coating, we will introduce charge attractions.
Funded by The Leverhulme Trust (42 months from April 2019)
Polymers are large molecules, typically synthesized from petroleum sources. The size of the polymer molecules has an effect on the adhesion to the surface and the temperature at which it softens. This project aims to answer the simple question: if a mixture of large and small linear polymers of the same composition are deposited from a solution to make a film, will they be randomly mixed, or will one of the chain populations be enriched at the surface? Polymers can also be synthesized in the shape of a star, with arms extending from a central core. We will also aim to answer the question: if a mixture of large and small star molecules (or linear and star polymers) is cast from a solution, will the small molecules be enriched at the surface? This project will provide excellent training for two undergraduates working closely with a post-doctoral researcher. To answer these questions, a student will model the non-equilibrium systems. In experiments, students will scatter high energy ions from the polymer films to determine their composition as a function of depth. The extent of layering or stratification will be precisely measured. The research will provide new understanding of ways to control surface properties of polymers.
Funded by the ACS Petroleum Research Fund (Sept. 2019 - August 2021)
Colloidal particles in liquids are found in many ordinary items, such as foods, paints, inks, cosmetics, and pharmaceuticals. Colloids in liquid are not static, but they are constantly moving in random directions because of Brownian diffusion. In this research, we will investigate ways to move colloids in water in specific desired directions.
When colloids have an electric charge they can be steered by an external electric field acting upon them through the mechanism known as electrophoresis. Recently, scientists have discovered that the electric fields created by salts dissolved non-uniformly in water can drive colloid motion. The colloidal particles will move up or down a gradient in salt concentration, depending on the type of salt and whether the charge on the particles is positive or negative.
Our vision in this project is to steer particles on curved paths by putting them in solutions in which there are gradients of two different salts going in directions at right angles to each other. We will use criss-crossed polymeric fibres to release salts into water as a way to devise complex patterns of salt concentrations. According to some recent calculations, it should be possible in these salt gradients to separate mixtures of particles that differ in their charge. This new concept will allow particles to be sorted in by a simple, low-cost method. An immediate application will be in re-using and recycling expensive colloids to minimise waste. The use of crossed gradients in salt concentration could also provide a way to measure the unknown electric charge on particles. The cross gradients of salt concentrations will provide a simple diagnostic method to identify particles, such as viruses or contaminants.
Having such precise control of colloid motion will open up possibilities for fabricating complex materials one particle at a time. We will attach some “sticky” molecules onto the colloids so that they will attach to surfaces after being steered their by electrophoresis. Our fundamental research might thereby lead to breakthroughs in the manufacturing of materials for applications in photonics, electronic devices or drug delivery.
Funded by EPSRC New Horizons Programme (February 2021 - November 2022)
The ‘Pulpex UK Bioscience Initiative’ will help Pulpex Ltd fully achieve its medium- term ambition by replacing its existing petrochemical-based barrier coatings with truly sustainable coatings as barriers to water and foods. The barrier coatings are applied on the interior of the Pulpex sustainable bottle. Sustainable materials are the future of food packaging; consumers, general public and corporates are demanding this product now.
Pulpex has already demonstrated the manufacture of a paper bottle that will enable brands to switch from glass or plastic to a sustainable alternative that is readily recycled. Pulpex has developed an advanced manufacturing process that converts cellulose fibre into a bottle that can be easily recycled through the existing and well-established paper collection schemes that are available throughout the UK.
Pulpex are partnering with two UK biomanufacturing firms. CuanTec is a world leading firm based in the European Centre for Marine Biotechnology in Oban who produce chitosan from waste material from shellfish using a proprietary fermentation process. Cellucomp is an established company based in Fife with deep expertise in the extraction of micro- fibrillated cellulose (MFC) from waste root vegetables using bioprocessing techniques. These are world-class companies based here the UK and seeking expanding markets for their products.
This ambitious collaborative project draws on scientific expertise from across the UK to accelerate the adoption of bioprocessing-derived barrier coatings for Pulpex’s breakthrough fibre bottles and other high barrier packaging applications. The incredible calibre of our partners is testament to the high standards we aspire to. The project offers deep industry and academic collaboration across the entire supply chain – converting non-food waste (e.g. shells from crustaceans and discards from root vegetables) into high value high barrier packaging that is market ready and validated. We see properly bioprocessing as essential to the development of the next generation of sustainable products.
The project aim is to deliver and accelerate commercial high barrier products – derived from the bioprocessing / fermentation of chitosan and MFC from vegetal waste streams – that can be effectively applied to Pulpex fibre bottles at scale and certified as suitable for liquid food use.
Funded by Innovate UK Biomanufacturing (Sept. 2023 - Aug. 2025)
Humans currently produce approximately 583 billion plastic bottles every year, with 38.5 million being used daily in the UK. Of this number in the UK, 39% are not recycled and 2% are littered, with devastating impact on soil, water and wildlife. A new approach to solve the problem of waste plastic packaging is offered by bottles made from wood pulp. In a process developed by Pulpex, wood-pulp bottles are sprayed internally with specialised coatings designed to be a barrier to prevent liquids and gases from seeping through the walls.
Pulpex bottles are made from sustainably sourced, FSC-certified wood pulp. Bottles can be recycled in the same way as paper and card. In the event that the bottles are not recycled or correctly disposed of, they will degrade readily in the natural environment. The carbon footprint for wood pulp bottles is 30% less than for PET.
Coatings inside the bottles are deposited from a wet mixture of different types of particles. The barrier properties of the coatings are easily affected by small changes in the drying conditions. This research will test the hypothesis that when the evaporation of water across a surface is non-uniform, pH gradients and particle flow will be induced within the coating, ultimately leading to a degradation of properties.
Royal Society Short Industry Fellowship (Oct. 2022 - Aug. 2023)
Approximately 38.5 million plastic bottles are used daily in the UK. Of this number, 39% are not recycled, and unfortunately 2% are littered, with devastating impact on soil, water and wildlife. Although individuals regret the environmental impact of plastics, and the high energy demand used in glass manufacturing, they have few alternatives when buying food and drink, cosmetics or homecare products.
To address this problem, Pulpex Ltd has demonstrated the manufacture of a paper bottle that will enable commercial brands to switch their packaging from glass or plastic to a sustainable alternative. Pulpex has developed an advanced manufacturing process that converts cellulose fibre into a bottle that can be easily recycled through existing, well-established paper collection schemes. Moreover, the carbon footprint for these paper bottles is 30% less than for PET. The Pulpex bottle presently uses a petrochemical-based coating to provide a physical barrier to contain liquids. Pulpex has a medium-term ambition to introduce truly sustainable, natural materials that will degrade harmlessly in the environment, if not recycled with paper.
Professor Keddie will apply his lifetime’s knowledge of coatings to develop sustainable barrier materials for the Pulpex bottle. He will focus on chitosan, a natural material derived from crustaceans. Chitosan will be modified so that liquids will permeate much more slowly through coatings made from it. Using physical chemistry principles, multiple layers will be engineered to make liquid flow less favourable. Ultimately, these coatings will be applied to Pulpex paper bottles at scale and certified for liquid food use.
Royal Society Industry Fellowship (Sept. 2024 - Aug. 2028)
Research collaborations
Our research is inter-disciplinary, and so we collaborate regularly with chemists, who synthesise molecules and particles, with microbiologists with expertise in bacteria, and with engineers with expertise in applications. Our work is appreciated and supported by industry.
- Previous EC-funded projects include
BarrierPlus: developed waterborne, surfactant-free barrier coatings for steel
NAPOLEON: produced waterborne polymer nanocomposites for applications in adhesives, coatings and cosmetics.
NSHAPE: designed core-shell, crosslinking polymer particles for pressure-sensitive adhesives
- Recent collaborations with resulting joint publications with
- Dr Cecile Dreiss (King's College London)
- Prof. Alex Routh (University of Cambridge)
- Prof. Jose Asua (POLYMAT, University of the Basque Country, UPV/EHU)
- Prof. Stefan Bon (University of Warwick)
- Prof. Alan Dalton (University of Sussex)
- Prof. Otto Muskens and Dr. Antonios Kanaras (University of Southampton)
- Prof. Elodie Bourgeat-Lami, Frank D'Agosto, and Muriel Lansalot (Univ. Lyon)
- Dr. Suzie Hingley-Wilson (University of Surrey)
- Current industrial collaborations with Avery Dennison and Syngenta.
- Previous industrial collaborations with Synthomer, BASF, DSM, Allnex, AkzoNobel, and International Paint.
Royal Society Industry Fellowship with a 50% secondment with Pulpex Ltd, developing sustainable packaging for liquids.
Indicators of esteem
2017 Thomas Graham Lecturer awarded by the RSC/SCI Joint Colloids Group
2015 Medallion from Coatings Societies International
Fellow of the Institute of Physics
International Scientific Advisory Committee of Advanced Research Center for Chemical Building Blocks in the Netherlands
Editorial Advisory Board of ACS Applied Materials and Interfaces
Awards Co-ordinator for the RSC/SCI Joint Colloids Group
Trustee of the High Polymer Research Group
2001 Winner of the Paterson Medal and Prize from the Institute of Physics for “major contributions to the understanding of the dynamics of polymers at surfaces, in thin films and in colloid dispersions”
Teaching
Presently, I am the module co-ordinator for Experimental and Professional Skills (ENG1091), offered to mechanical engineering students.
Previously, I developed and co-ordinated a module on Properties of Matter (PHY1039), which introduces first-year students to classical thermodynamics and solid state physics.
I was previously the Co-ordinator of the Year One practical laboratory on "Properties of Matter" and "Atoms and Quanta."
I also developed and co-ordinated a module on Soft Matter and Biological Physics (PHY3040). I delivered lectures to final-year undergraduates on molecular interactions, phase equilibria, dynamics of liquid, colloids, and polymer physics.
I am the Senior Tutor for the Professional Training work placement programme for the Department of Physics. Typically, up to 20 students spend a year working in industry each year. I manage their assessment, and I support students preparing for a placement year.
Sustainable development goals
My research interests are related to the following:



Publications
Highlights
- Our paper in Physical Review Letters on "Dynamic Stratification in Drying Films of Colloidal Mixtures," has an Altmetric score of 689 (in the 99th percentile for publications of a similar age). We report our discovery a mechanism for stratification in which small colloidal particles accumulate on a layer of larger particles.
- Our experimental and computational work to test a model of stratification in colloidal films, entitled "Stratification in binary colloidal polymer films: experiment and simulations", is freely available to read in the RSC journal, Soft Matter. We gain a deeper understanding of the parameters that result in stratification.
- We have reported a method to assembly plasmonic nanoparticles in a periodic structure templated by colloids in a publication in ACS Nano entitled "Fast Assembly of Gold Nanoparticles in Large-Area 2D Nanogrids Using a One-Step, Near-Infrared Radiation-Assisted Evaporation Process." It is freely available to read.
- I have co-authored a book entitled Fundamentals of Latex Film Formation: Processes and Properties, published by Springer in 2010.
Biocoatings are typically colloidal polymer films confining metabolically active, nongrowing bacteria. Depending on the species of confined bacteria, biocoatings find applications in wastewater treatment, biofuel production, carbon fixation, environmental remediation, biosensing, and more. However, the successful use of biocoatings faces numerous challenges, including a low permeability to reactants and metabolized products, osmotic stress on bacteria during drying of the coatings, and cell dehydration leading to bacteria death. Here, we address these challenges through two interlinked processing methods. (1) Coagulant gelation of the colloidal polymer dispersion creates a porous microstructure with high permeability. (2) Wet sintering by immersion in a liquid medium reduces osmotic stress and avoids desiccation of the bacteria. In a model system of Escherichia coli in an acrylic copolymer latex biocoating, these two methods yielded a cell viability that is approximately 500 times greater than the conventional method of biocoating formation using dry sintering in air at an elevated temperature. We have discovered that when lysogeny broth is used as the medium for wet sintering, the cell viability is significantly higher than that for other liquids. Increasing the salt concentration for coagulant gelation leads to thicker coatings (and hence more cells per area of the coating). However, cell viability decreases when the salt concentration is increased, so a compromise is needed. Metabolic activity of E. coli in a wet-sintered biocoating was demonstrated through the production of ethanol as a biofuel. These results hold promise for the future exploitation of biocoatings using a broad range of bacterial, especially desiccation-intolerant species.
Environmental regulations and consumer demand are driving the need for packaging materials to reduce or eliminate the use of petroleum-based plastics by replacing them with natural and degradable alternatives. Water flux through a barrier layer is governed by the differential in water activity between a high side (e.g., an aqueous solution) and a low side, such as humid air. We propose that a water-absorbent layer sandwiched between two barrier layers will act as a sponge and locally raise the water activity. It will thereby lower the activity differential across the first barrier layer and, hence, reduce the water flux through the multilayer for a set time period. We present a theoretical model that predicts the water flux through a multilayered structure of two or more barrier layers sandwiching hydrophilic layers that hold water according to an absorption isotherm. We use this model to evaluate the effects of layer thicknesses and distributions, the barrier permeability, the water-holding capacity, and initial water activity of the absorbent layers. We found that the water mass loss rate decreases when the thickness and water-holding capacity of the absorbent layer increase. The greatest reduction in the mass loss through a multilayer was achieved when the absorbent layer had an initial water activity of 0 (fully dried). The relative thicknesses of the barrier layers in the multilayer also have an impact on the water loss rate; thicker barrier layers on the low-activity side of the multilayer are the most effective in reducing the water flux. We have verified the model in an ideal system. We also investigated a multilayer structure of a waterborne emulsion polymer barrier coating and water-absorbing chitosan, which is a deacetylated form of chitin. Here, the chitosan layer offered little benefit in decreasing the water permeability because the layers were not thick enough and the permeability of the waterborne coating on its own is not sufficiently low. With our design concept for multilayer barriers containing absorbent layers and the underpinning theoretical model, we envisage materials systems with enhanced barrier properties while also using less petrochemically derived plastic.
•Thin, maltodextrin films were decorated with ink-jet printed insoluble cocoa butter patterns.•The heterogeneity Area Coverage (AC) affects the spreading dynamics of water droplets.•Area Coverage of 8% doubles the spreading time, while AC 26 % trebles it. AC < 1 % shows no significant effect.•Heterogeneities can affect wetting more than the molecular mass of the soluble substrate.•These results suggest surface design criteria to quantitatively control wetting. Recent studies have highlighted the complex mechanisms governing the spreading of a solvent onto a homogeneous soluble film, such as water on soluble polysaccharides. The presence of surface fat slows down the wetting of spray dried food powders, but this phenomenon is not yet understood quantitatively. In this study, surface heterogeneities were created by the ink-jet printing of cocoa butter onto water-soluble maltodextrin thin films to produce hydrophobic deposits with a range of area coverages. The spreading dynamics of water was studied controlling relative humidity. Area coverages above 1.2 % were found to decrease the contact line speed and increase the contact angle. The contact line was deformed by the deposits of cocoa butter, causing in some cases periodic decelerations followed by accelerations. Area coverages above 26 % led to a three-fold increase in the spreading time. These novel insights could help to design soluble heterogeneous surfaces meeting a desired wetting performance.
With the increasing use of pressure-sensitive adhesives (PSAs) in various industries, there is a need for greater sustainability, particularly in developing polymer materials from renewable resources, as well as the reuse and recycling of materials to reduce environmental impact, reduce waste, or extend their life. Here, we outlined the required properties of PSAs which are governed by the molecular parameters (molecular weights, dispersities, molecular wight between entanglement, molecular weight between cross-links and gel content) of polymer materials which subsequently define the physical properties (storage and loss moduli, glass transition temperature) that are required for good performance in peel, tack and shear tests. The sustainable approach discussed here is the development of degradable polymer materials featuring selectively degradable linkages in the backbone. This provides a viable alternative for the design of PSAs that could overcome the 'stickies' problem and make the recycling of glass and cardboard more efficient.
Interest in Pickering emulsions, which are stabilized by nanoparticles, has been driven by their superior stability and a desire to avoid the use of conventional surfactants. However, understanding of the chemical and physical phenomena governing particle stabilization at liquid/liquid interfaces remains limited because of the complexity of these systems. In particular, discrepancies can emerge between the inherent thermodynamic and the observed three-phase contact angles in such systems (particle/oil/water). We address this issue by modifying the classic equation for the free energy of a spherical nanoparticle at an oil/water interface. Our model defines the range of three-phase contact angles that enable successful Pickering stabilization. The model shows that the highest destabilization energy occurs when equals the position angle , rather than 90°, as found in the conventional model. Our findings have significant implications for the identification of candidate Pickering stabilizers and the design of the emulsification process.
In an externally imposed electrolyte (salt) concentration gradient, charged colloids drift at speeds of order one micrometre per second. This phenomenon is known as diffusiophoresis. In systems with multiple salts and 'crossed' salt gradients, a nonlocal component of the electric field associated with a circulating (solenoidal) ion current can arise. This is in addition to the conventional local component that depends only on the local salt gradients. Here we report experimental observations verifying the existence of this nonlocal contribution. To our knowledge this is the first observation of nonlocal diffusiophoresis. The current develops quasi-instantaneously on the time scale of salt diffusion. Therefore, in systems with multiple salts and crossed salt gradients, one can expect a nonlocal contribution to diffusiophoresis which is dependent on the geometry of the system as a whole and appears as a kind of instantaneous 'action-at-a-distance' effect. The interpretation is aided by a magnetostatic analogy. Our experiments are facilitated by a judicious particle-dependent choice of salt (potassium acetate) for which the two local contributions to diffusiophoresis almost cancel, effectively eliminating conventional diffusiophoresis. This enables us to clearly identify the novel, nonlocal effect and may be useful in other contexts, for example in sorting particle mixtures.
Waterborne colloidal polymer coatings are widely used in architectural and agricultural applications where they are subject to challenging environments, such as extremes of temperatures and relative humidities (RH). This research investigates the effects of adding two common co-formulants, poly(acrylic acid) (PAA) and xanthan gum (XG), to waterborne polymer composite coatings in these environments. The mechanical properties of the resulting coatings are of particular interest. Hardness, creep and tack properties of thick (similar to 400 mu m) formulated model coatings were characterized using a micro-indentation technique operating in a single cycle within a bespoke environmental chamber. Measurements were made at three temperatures (16, 20 and 30 degrees C), which span the glass transition temperature (T-g) of the acrylic copolymer binder, and over three RH values of 10%, 43%, and 90%. The creep data were analysed using the Burgers model to extract characteristic viscoelastic properties. The tack was found by recording the force when withdrawing the probe from the sample and using it to obtain nominal stress (knowing the indentation depth and probe geometry) during the indenter's withdrawal and hence the work of adhesion (W-Adh) to detach from the coating. Tack adhesion is completely lost below the binder's T-g but increases when the ambient temperature increases. In formulated coatings, both the tack and creep deformation increase as the relative humidity increases, and this trend is observed at each temperature. There is no evidence from thermal analysis for plasticization of the acrylic polymer by moisture sorption, but the two co-formulants are hydrophilic. The observed softening of the coatings at high RH can be attributed to water sorption in the components. The presence of glassy PAA has the effect of raising the hardness of glassy coatings, but only at low RH when there is no plasticization by water. The addition of hydrophilic XG surprisingly reduces tack adhesion while also raising the viscosity of the coating. These findings will inform the formulation of waterborne colloidal coatings to function in a range of environments.
The uses of pressure-sensitive adhesives (PSAs) are wide ranging, with applications including labels, tapes, and graphics. To achieve good adhesion, a PSA must exhibit a balance of viscous and elastic properties. Previous research has found that a thin, elastic surface layer on top of a softer, dissipative layer resulted in greater tack adhesion compared with the single layers. Superior properties were achieved through a bilayer obtained via successive depositions, which consume energy and time. To achieve a multilayered structure via a single deposition process, we have stratified mixtures of waterborne colloidal polymer particles with two different sizes: large poly(acrylate) adhesive particles (ca. 660 nm in diameter) and small poly(butyl acrylate) (pBA) particles (ca. 100 nm). We used two types of pBA within the particles: either viscoelastic pBA without an added cross-linker or elastic pBA with a fully cross-linked network. Stratified surface layers of deuterium-labeled pBA particles with thicknesses of at least 1 μm were found via elastic recoil detection and qualitatively verified via the analysis of surface topography. The extent of stratification increased with the evaporation rate; films that were dried slowest exhibited no stratification. This result is consistent with a model of diffusiophoresis. When the elastic, cross-linked pBA particles were stratified at the surface, the tack adhesion properties made a transition from brittle failure to tacky. For pBA without an added cross-linker, all adhesives showed fibrillation during debonding, but the extent of fibrillation increased when the films were stratified. These results demonstrate that the PSA structure can be controlled through the processing conditions to achieve enhanced properties. This research will aid the future development of layered or graded single-deposition PSAs with designed adhesive properties.
High quality opal-like photonic crystals containing graphene are fabricated using evaporation-driven self-assembly of soft polymer colloids. A miniscule amount of pristine graphene within a colloidal crystal lattice results in the formation of colloidal crystals with a strong angle-dependent structural color and a stop band that can be reversibly shifted across the visible spectrum. The crystals can be mechanically deformed or can reversibly change color as a function of their temperature, hence their sensitive mechanochromic and thermochromic response make them attractive candidates for a wide range of visual sensing applications. In particular, we show that the crystals are excellent candidates for visual strain sensors or integrated time-temperature indicators which act over large temperature windows. Given the versatility of these crystals, this method represents a simple, inexpensive and scalable approach to produce multifunctional graphene infused synthetic opals and opens up exciting applications for novel solution-processable nanomaterial based photonics.
The film formation of an acrylate latex with a glass-transition temperature of 38 °C has been achieved through the use of near-infrared (NIR) radiative heating. A hard, crack-free coating was obtained without the addition of plasticizers. Sintering of acrylate particles was confirmed through measurements using atomic force microscopy. The addition of an NIR-absorbing polymer increased the rate of particle deformation such that it was significantly greater than obtained in a convection oven at 60 °C. The results are consistent with a lower polymer viscosity under infrared radiation, according to a simple analysis using a standard model of sintering.
When fabricating photonic crystals from suspensions in volatile liquids using the horizontal deposition method, the conventional approach is to evaporate slowly to increase the time for particles to settle in an ordered, periodic close-packed structure. Here, we show that the greatest ordering of 10 nm aqueous gold nanoparticles (AuNPs) in a template of larger spherical polymer particles (mean diameter of 338 nm) is achieved with very fast water evaporation rates obtained with near-infrared radiative heating. Fabrication of arrays over areas of a few cm2 takes only seven minutes. The assembly process requires that the evaporation rate is fast relative to the particles’ Brownian diffusion. Then a two-dimensional colloidal crystal forms at the falling surface, which acts as a sieve through which the AuNPs pass, according to our Langevin dynamics computer simulations. With sufficiently fast evaporation rates, we create a hybrid structure consisting of a two-dimensional AuNP nanoarray (or “nanogrid”) on top of a three-dimensional polymer opal. The process is simple, fast and one-step. The interplay between the optical response of the plasmonic Au nanoarray and the microstructuring of the photonic opal results in unusual optical spectra with two extinction peaks, which are analyzed via finite-difference time-domain method simulations. Comparison between experimental and modelling results reveals a strong interplay of plasmonic modes and collective photonic effects, including the formation of a high-order stop band and slow-light enhanced plasmonic absorption. The structures, and hence their optical signatures, are tuned by adjusting the evaporation rate via the infrared power density.
1 figure.-- Talk delivered at the "Designed Assembly of Colloids at Interfaces - Fundamentals to Applications. Online event", organised by the Liquids and Complex Fluids Group, Institute of Physics (Great Britain), 14th-16th June 2021. In biomedical or pharmaceutical applications, surfactants in conventional emulsions can cause tissue irritation or cell damage. In this work, biocompatible nanoparticles of sp3 carbon, called nanodiamond (ND), with a carboxylated surface, were used to prepare mono-sized Pickering emulsions of sunflower oil in water. Emulsification of the oil in acidic ND dispersions was achieved via two different techniques: membrane emulsification and probe sonication. Whereas membrane emulsification fabricated larger (about 30 μm) oil droplets (Figure 1), probe sonication resulted in smaller (sub-μm) droplets and higher stability for a wide range of pH values (acidic to alkaline). The emulsions show high stability against mechanical vibration and offer long-term storage up to one year. Pendant drop tensiometry confirmed that ND particles are adsorbed at the oil/water interface, with a greater decrease in interfacial tension found with increasing ND concentrations in the dispersion. ND coatings become more hydrophilic with increasing pH, according to three-phase contact angle analysis, because of deprotonation of the carboxylic acid groups. Destabilisation of the Pickering emulsion at very high pH was demonstrated in highly alkaline buffers, which is due to the saponification of the vegetable oil and association with cations in solution. This is the first systematic study of carboxylic ND-stabilized Pickering emulsions, and it indicates a very promising prospect for a range of applications.
Bacteria are used in a range of sectors, such as wastewater treatment, bioremediation, or as soil additives. For these applications, live bacteria are encapsulated to protect them from mechanical damage and desiccation. Unlike other types of cargo, bacteria are not always required to be released because when encapsulated, they can interface with their environment and fulfill their roles via molecular transport through the capsule walls. The aims of encapsulation are then shifted away from delaying release to making capsules that are mechanically robust while permitting sufficient diffusion to support the metabolic activity of the bacteria. Here, we produced covalent hydrogel capsules from a water-in-oil (W/O) emulsion of aqueous poly(ethylene glycol) diacrylate (PEGDA) in hexadecane containing a UV-radical initiator. Upon initiation, PEGDA polymerization begins at the W/O interface to produce hydrogel capsules. We discovered three classes of capsule microstructures with differing levels of macroporosity that could be tailored by changing the polymerization conditions. Systematic investigations showed how the UV energy input and the PEGDA macromonomer concentration can be used to selectively create honeycomb, sponge like, or dense spherical capsules. To explain the sponge-like structure, we propose a capsule formation mechanism based on diffusion-limited aggregation of PEGDA microbeads. The structures resemble random-walk simulations of sticky beads and, furthermore, satisfy the theoretical volume fractions required for percolation. We successfully encapsulated live Mycobacterium smegmatis within the sponge structures, demonstrating biocompatibility. Importantly, the internal hydrogel microstructure allows the growth of bacteria. This mechanistic understanding is paramount for designing robust covalent capsules while optimizing porosity within hydrogel structures.
The distribution of surfactants in waterborne colloidal polymer films is of significant interest for scientific understanding and defining surface properties in applications including pressure-sensitive adhesives and coatings. Because of negative effects on appearance, wetting, and adhesion, it is desirable to prevent surfactant accumulation at film surfaces. The effect of particle deformation on surfactant migration during film formation was previously investigated by Gromer et al. through simulations, but experimental investigations are lacking. Here, we study deuterium-labeled sodium dodecyl sulfate surfactant in a poly(butyl acrylate) latex model system. The particle deformability was varied via cross-linking of the intraparticle polymer chains by differing extents. The cross-linker concentration varied from 0 to 35 mol % in the copolymer, leading to a transition from viscoelastic to elastic. Ion beam analysis was used to probe the dry films and provide information on the near-surface depth distribution of surfactant. Films of nondeformable particles, containing the highest concentration of cross-linker, show no surfactant accumulation at the top surface. Films from particles partially deformed by capillary action show a distinct surfactant surface layer (ca. 150 nm thick). Films of coalesced particles, containing little or no cross-linker, show a very small amount of surfactant on the surface (ca. 20 nm thick). The observed results are explained by considering the effect of cross-linking on rubber elasticity and applying the viscous particle deformation model by Gromer et al. to elastically deformed particles. We find that partially deformed particles allow surfactant transport to the surface during film formation, whereas there is far less transport when skin formation acts as a barrier. With elastic particles, the surfactant is carried in the water phase as it falls beneath the surface of packed particles. The ability to exert control over surfactant distribution in waterborne colloidal films will aid in the design of new high-performance adhesives and coatings.
The properties of spin-on titanium-nitride (SO-TiN) thin films were optimized for integrated- circuit application. The two steps of the spin-on process were characterized: one, the initial step in which a thin-film titanium oxide is formed, and two, the conversion of the thin film to titanium-nitride (TiN) by rapid thermal processing in ammonia. The spin-on TiN showed a uniform coating on a flat wafer surface for all the precursors. However, on non-planar topography some solutions produced cracked films while others did not. The precursor's effect was investigated, and it is proposed that the optimized precursor should include more carbon in the initial annealing stage so the film does not crack. The chosen precursor, titanium- tertiary-butoxide, was investigated and characterized versus processing temperature, heating rate, and gas flow. The experiment was designed at the 700 degree(s)C - 1000 degree(s)C temperature range, with 0.1 - 200 degree(s)C/sec. heating rate, and hold time of 30 - 300 sec. at the upper temperature before rapid cool down. The optimal processing conditions at NH3 are heating at 100 - 120 degree(s)C/sec. ramp from room temperature up to 900 degree(s)C - 1000 degree(s)C where the wafer is annealed for 30 - 100 seconds before rapid cool down to room temperature.
Pressure-sensitive adhesives (PSAs) are made from soft, irreversibly lightly crosslinked polymers. Even after removal from surfaces, they retain insoluble networks which pose problems during the recycling of glass and cardboard. Herein, degradable PSAs are presented that provide the required performance in use but have networks that can be degraded after use. A series of copolymers was prepared through radical copolymerization of n-butyl acrylate, 4-acryloyloxy benzophenone (ABP) photo-crosslinker, and dibenzo[c,e]oxepin-5(7H)-thione (DOT) to provide degradable backbone thioesters. The optimum tack and peel strengths were found for molar contents of 0.05 mol% ABP and 0.25 mol% DOT. Degradation of the backbone thioesters through aminolysis or thiolysis led to the full dissolution of the networks, loss of adhesive properties of films (decreases in the measured tack and peel strengths), and the quick detachment of model labels from a substrate. Inclusion of DOT into PSAs offers a viable route toward degradable and recyclable packaging labels.
We highlight the significance of capillary pressure in the directed assembly of nanorods in ordered arrays of colloidal particles. Specifically, we discuss mechanisms for the assembly of carbon nanotubes at the interstitial sites between latex polymer particles during composite film formation. Our study points to general design rules to be considered to optimize the ordering of nanostructures within such polymer matrices. In particular, gaining an understanding of the role of capillary forces is critical. Using a combination of electron microscopy and atomic force microscopy, we show that the capillary forces acting on the latex particles during the drying process are sufficient to bend carbon nanotubes. The extent of bending depends on the flexural rigidity of the carbon nanotubes and whether or not they are present as bundled ensembles. We also show that in order to achieve long-range ordering of the nanotubes templated by the polymer matrix, it is necessary for the polymer to be sufficiently mobile to ensure that the nanotubes are frozen into the ordered network when the film is formed and the capillary forces are no longer dominant. In our system, the polymer is plasticized by the addition of surfactant, so that it is sufficiently mobile at room temperature. Interestingly, the carbon nanotubes effectively act as localized pressure sensors, and as such, the study agrees well with previous theoretical predictions calculating the magnitude of capillary forces during latex film formation.
The loss of optical transparency when polymer films are immersed in water, which is called “water whitening,” severely limits their use as clear barrier coatings. It is found that this problem is particularly acute in films deposited from polymers synthesized via emulsion polymerization using surfactants. Water whitening is less severe in secondary dispersion polymers, which are made by dispersing solution polymers in water without the use of surfactants. NMR relaxometry in combination with optical transmission analysis and electron microscopy reveal that some of the water sorbed in emulsion polymer films is contained within nano-sized “pockets” or bubbles that scatter light. In contrast, the water in secondary dispersion polymer films is mainly confined at particle interfaces, where it scatters light less strongly and its molecular mobility is reduced. The addition of surfactant to a secondary dispersion creates a periodic structure that displays a stop band in the optical transmission. The total amount of sorbed water is not a good indicator of polymers prone to water whitening. Instead, the particular locations of the water within the film must be considered. Both the amount of water and the size of the local water regions (as are probed by NMR relaxometry) are found to determine water whitening.
Existing inorganic materials for radiation sensors suffer from several drawbacks, including their inability to cover large curved areas, lack of tissue equivalence toxicity, and mechanical inflexibility. As an alternative to inorganics, poly(triarylamine) (PTAA) diodes have been evaluated for their suitability for detecting radiation via the direct creation of X-ray induced photocurrents. A single layer of PTAA is deposited on indium tin oxide (ITO) substrates, with top electrodes selected from Al, Au, Ni, and Pd. The choice of metal electrode has a pronounced effect on the performance of the device; there is a direct correlation between the diode rectification factor and the metal-PTAA barrier height. A diode with an Al contact shows the highest quality of rectifying junction, and it produces a high X-ray photocurrent (several nA) that is stable during continuous exposure to 50 kV Mo K alpha X-radiation over long time scales, combined with a high signal-to-noise ratio with fast response times of less than 0.25 s. Diodes with a low band gap, 'Ohmic' contact, such as ITO/PTAA/Au, show a slow transient response. This result can be explained by the build-up of space charge at the metal-PTAA interface, caused by a high level of charge injection due to X-ray-induced carriers. These data provide new insights into the optimum selection of metals for Schottky contacts on organic materials, with wider applications in light sensors and photovoltaic devices.
A new application of infrared ellipsometry is reported. Specifically, the interdiffusion between thin films of miscible polymers - poly(methyl methacrylate) and poly(vinylidene fluoride) - is detected in a non-invasive measurement. A novel technique of data analysis for interdiffusion was developed and is described. The validity of the approach is supported by simulations of diffusion in a bilayer. The onset of extensive interdiffusion over a time period of 15 min occurs at a temperature of 160°C. At a temperature of 190°C, the data show that complete mixing of a bilayer (850 nm thick) occurs within 30s, which is consistent with previously reported values of the mutual diffusion coefficient. Infrared ellipsometry is non-invasive, applicable at elevated temperatures and relatively fast and sensitive. Although, in these measurements, it was unable to determine a concentration profile at the interface, infrared ellipsometry was used successfully to detect when interdiffusion had occurred. Hence, it is a useful means for screening polymer pairs for miscibility. Copyright © 2004 John Wiley & Sons, Ltd.
Implantable microelectrodes have the potential to become part of neural prostheses to restore lost nerve function after nerve damage. The initial adsorption of proteins to materials for implantable microelectrodes is an important factor in determining the longevity and stability of the implant. Once an implant is in the body, protein adsorption takes place almost instantly before the cells reach the surface of an implant. The aim of this study was to identify an optimum material for electrode recording sites on implantable microelectrodes. Common materials for electrode sites are gold, platinum, iridium, and indium tin oxide. These, along with a reference material (titanium), were investigated. The thickness and the structure of adsorbed proteins on these materials were measured using a combination of atomic force microscopy and ellipsometry. The adsorbed protein layers on gold (after 7 and 28 days of exposure to serum) were the smoothest and the thinnest compared to all the other substrate materials, indicating that gold is the material of choice for electrode recording sites on implantable microelectrodes. However, the results also show that indium tin oxide might also be a good choice for these applications.
A new class of X-ray sensor – in which there is a blend of poly(triarylamine) (PTAA) and 6,13-bis(triisopropylsilylethynyl) (TIPS)-pentacene in the active layer of a diode structure – has been developed. The crystalline pentacene provides a fast route for charge carriers and leads to enhanced performance of the sensor. The first time-of-flight charge-carrier mobility measurement of this blend is reported. The mobility of PTAA and TIPS-pentacene in a 1:25 molar ratio was found to be 2.2 × 10−5 cm2 V−1 s−1 (averaged for field strengths between 3 × 104 and 4 × 105 V cm−1), which is about 17 times higher than that obtained in PTAA over the same range of field strengths. This higher mobility is correlated with a fourfold increase in the X-ray detection sensitivity in the PTAA:TIPS-pentacene devices.
The isothermal structural relaxation of glassy, spin-cast polymer thin films has been investigated. Specifically, the thickness h of freshly cast poly(methyl methacrylate) thin films was measured over time using spectroscopic ellipsometry. The spin-cast films exhibit a gradual decrease in thickness, which is attributed to structural relaxation of the glass combined with simultaneous solvent loss. In all cases, It was found to be greater than the equilibrium thickness h(infinity), which is obtained by cooling slowly from the melt. It is observed that both the rate of the volume relaxation and the fractional departure from h. (referred to as delta(0)) increase with increasing film thickness. In the limit of very thin films, the initial h is close to h(infinity), and delta(0) is small, whereas in thick films (>500 nm), a plateau value of delta(0) of 0.16 is observed, which is close to the volume fraction of the solvent at the vitrification point. This dependence of) on thickness is observed regardless of the substrate, polymer molecular weight, or angular velocity during spin casting. Enhanced mobility near film surfaces could be leading to greater relaxation in thinner films prior to, and immediately after, the vitrification of the polymer during the deposition process.
When films are deposited from mixtures of colloidal particles of two different sizes, a diverse range of functional structures can result. One structure of particular interest is a stratified film in which the top surface layer has a composition different than in the interior. Here, we explore the conditions under which a stratified layer of small particles develops spontaneously in a colloidal film that is cast from a binary mixture of small and large polymer particles that are suspended in water. A recent model, which considers the cross-interaction between the large and small particles (Zhou et al., Phys. Rev. Lett. (2017) 118, 108002), predicts that stratification will develop from dilute binary mixtures when the particle size ratio (), initial volume fraction of small particles ( s), and Péclet number are high. In experiments and Langevin dynamics simulations, we systematically vary and s in both dilute and concentrated suspensions. We find that stratified films develop when s is increased, which is in agreement with the model. In dilute suspensions, there is reasonable agreement between the experiments and the Zhou et al. model. In concentrated suspensions, stratification occurs in experiments only for the higher size ratio = 7. Simulations using a high Péclet number, additionally find stratification with = 2, when s is high enough. Our results provide a quantitative understanding of the conditions under which stratified colloidal films assemble. Our research has relevance for the design of coatings with targeted optical and mechanical properties at their surface.
It is well established that colloidal polymer particles can be used to create organised structures by methods of horizontal deposition, vertical deposition, spin-casting, and surface pattern-assisted deposition. Each particle acts as a building block in the structure. This paper reviews how two-phase (or hybrid) polymer colloids can offer an attractive method to create nanocomposites. Structure in the composite can be controlled at the nanoscale by using such particles. Methods to create armored particles, such as via methods of hetero-flocculation and Pickering polymerization, are of particular interest here. Polymer colloids can also be blended with other types of nanoparticles, e.g. nanotubes and clay platelets, to create nanocomposites. Structure can be controlled over length scales approaching the macroscopic through the assembly of hybrid particles or particle blends via any of the various deposition methods. Colloidal nanocomposites can offer unprecedented long-range 2D or 3D order that provides a periodic modulation of physical properties. They can also be employed as porous templates for further nanomaterial fabrication. Challenges in the design and control of the macroscopic properties, especially mechanical, are considered. The importance of the internal interfacial structure (e.g. between inorganic and polymer particles) is highlighted.
The effects of the CTA concentration on polymerization kinetics, polymer microstructure, particle morphology, and adhesive performance of waterborne hybrid PSAs prepared by simultaneous free-radical and addition miniemulsion polymerizations are studied. The development of the microstructure is shown to differ from waterborne acrylic PSAs obtained by free-radical polymerization because of the contribution of the addition reaction, which in turn causes marked differences in the adhesive performance of the final films. A computer simulation is developed to obtain detailed information about the microstructure of PU/acrylic hybrids and to correlate the microstructure with the final adhesive properties. Copyright © 2013 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Reiter has recently reported a situation in which the dewetting of quasi-solid films is linked to plastic deformation--rather than viscous flow--resulting from capillary forces. Herein we propose that, in thin films of some glassy polymers--especially poly(methyl methacrylate) (PMMA)--prepared by spin-casting from solvent, structural relaxation might impart sufficient stress to cause plastic deformation. We find that PMMA films decrease in thickness by several percent, which is sufficient to create significant stress in those cases in which the film is attached to a rigid substrate. The floating technique, which can take tens of minutes, might allow most of the structural relaxation to occur prior to dewetting experiments.
Biofilm bioreactors are attracting growing interest in the wastewater industry, as they allow higher cell densities and thus higher reaction rates compared to conventional bioreactors. However, some commonly used nitrifying bacteria, such as Nitrosomonas europaea, are slow-growing and need a prolonged period of time to develop a mature biofilm. Here, a biocoating or "living paint" is introduced, which is a synthetic biofilm made from a colloidal polymer (synthetic latex) binder encapsulating viable nitrifying bacteria at high density. Conventionally, the film formation of biocoatings is achieved by drying a bacteria/latex mixture. However, this fabrication is detrimental to the viability of the encapsulated bacteria because of the osmotic stress induced by desiccation. A nondesiccating film formation process is presented for biocoatings, which exploits two colloid science phenomena: coagulation and wet sintering. Desiccation-sensitive, nitrifying bacteria are employed in the biocoatings to convert NH4+ to NO2- and then NO3-. These biocoatings have a conversion rate (NO2- and NO3- production) of 3 mg N g(-1) d(-1) that is five times higher than in conventionally desiccated biocoatings. The reactivity continues over a period of 1 month. The processing method for these living paints is transformative for wastewater treatment and other applications using delicate, desiccation-sensitive microorganisms.
Data obtained from spectroscopy ellipsometry: Film thickness as a function of temperature from dynamic and step-by-step scans. Determination of the coefficients of thermal expansion coefficients for a selection of 10 polymers from ellipsometry data and PVT data.
Nanoparticles of cerium dioxide (or nanoceria) are of interest because of their oxygen buffering, photocatalytic ability, and high UV absorption. For applications, the nanoceria can be incorporated in a polymer binder, but questions remain about the link between the nanoparticle distribution and the resulting nanocomposite properties. Here, the thermal, mechanical and optical properties of polymer/ceria nanocomposites are correlated with their nanostructures. Specifically, nanocomposites made from waterborne Pickering particles with nanoceria shells are compared to nanocomposites made from blending the equivalent surfactant-free copolymer particles with nanoceria. Two types of nanoceria (protonated or citric acid-coated) are compared in the Pickering particles. A higher surface coverage is obtained with the protonated ceria, which results in a distinct cellular structure with nanoceria walls within the nanocomposite. In the blend of particles, a strong attraction between the protonated nanoceria and the acrylic acid groups of the copolymer likewise leads to a cellular structure. This structure offers transparency in the visible region combined with strong UV absorption, which is desired for UV blocking coating applications. Not having an attraction to the polymer, the citric acid-coated nanoceria forms agglomerates that lead to undesirable light scattering in the nanocomposite and yellowing. This latter type of nanocomposite coating is less effective in protecting substrates from UV damage but provides a better barrier to water. This work shows how the nanoparticle chemical functionalization can be used to manipulate the structure and to tailor the properties of UV-absorbing barrier coatings.
A significant reduction in the electrical percolation threshold is achieved by locking carbon nanotubes (CNTs) in a predominantly hexagonally close-packed (HCP) colloidal crystal lattice of partially plasticized latex particles. Contrary to other widely used latex processing where CNTs are randomly distributed within the latex matrix, for the first time, we show that excluding CNTs from occupying the interior volume of the latex particles promotes the formation of a nonrandom segregated network. The electrical percolation threshold is four times lower in an ordered segregated network made with colloidal particles near their glass transition temperature (T(g)) in comparison to in a random network made with particles at a temperature well above the T(g). This method allows for a highly reproducible way to fabricate robust, stretchable, and electrically conducting thin films with significantly improved transparency and lattice percolation at a very low CNT inclusion which may find applications in flexible and stretchable electronics as well as other stretchable technologies. For instance, our technology is particularly apt for touch screen applications, where one needs homogeneous distribution of the conductive filler throughout the matrix.
Previous theoretical and experimental work has shown that surface tension gradients in liquid layers create surface defects and inhibit the levelling of an uneven surface. In coatings deposited from thermosetting polyester powders, which are studied here, small amounts of a low molecular-weight acrylate are incorporated to act as a “flow agent.” We find that this additive lowers the surface tension of the polymer melt and has a minor effect on the melt viscosity. A slower rate of levelling results from the decreased surface tension. We provide experimental evidence that lateral gradients in the surface tension of the polymer melt, resulting from the non-uniform distribution of the flow agent, inhibit the levelling of the surface. Specifically, the surface roughness of a powder coating is up to three times greater when a steep surface tension gradient is purposely created through powder blending. Surface tension gradients might also be responsible for the greater surface roughness (observed with atomic force microscopy on lateral length scales of 100 μm) that is found in coatings that contain flow agent.
Excimer laser irradiation is used to crystallize hydrogenated amorphous silicon thin films. The resulting films show a stratified microstructure with a crystalline volume fraction of up to 90%. There is a range of excimer laser energy that can produce stratified nanocrystalline silicon with a Tauc gap as high as 2.2 eV. This value is greater than that of amorphous or crystalline silicon and is contrary to that predicted from the theoretical analysis of mixed-phase silicon thin films. The phenomenon is explained by employing transmission electron microscopy and spectroscopic ellipsometry, and the observed bandgap enhancement is associated with quantum confinement effects within the nanocrystalline silicon layers, rather than an impurity variation.
Colloidal nanocomposite adhesives are made by blending soft adhesive particles with hard nanoparticles (NPs) that sit at the particle boundaries to create a percolating phase. When the nanocomposite is heated with infrared (IR) radiation, the NPs sinter together to create a rigid structure that hardens the composite and thereby switches off the tack adhesion. It is discovered that the IR power density of an irradiation for 20 s must exceed a threshold value of 1.07 W cm before the tack is switched off. At lower power densities, an analysis of the sintering of the NPs shows that there is not sufficient time to link them together into a rigid structure. These results reveal that the switching of colloidal nanocomposite adhesives can be easily controlled through the IR power density and the time of the exposure. © 2014 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
The morphology of pressure sensitive adhesives (PSAs), especially at the surface in contact with a release liner, is expected to have a dominant influence on the tack strength and energy in an application. We have used tapping-mode atomic force microscopy to determine the morphology at the surfaces of freshly-cast waterborne acrylic PSAs over lateral length scales of a few m. We demonstrate that topographical features on silicone release liners can be used to pattern the PSA surface in contact with it. Control of the texture of a PSA surface can potentially be exploited to tailor its properties. Latex particle boundaries are much better defined at the air surface of the PSA in comparison to its back face. A series of experiments suggests that this difference results from the distribution of water-soluble species within the dry film. The pressures and processes involved in the transfer lamination process do not alter the PSA morphology. The first reported AFM images of the response of these materials to pressure and shear provide insight into the deformation mechanisms. Amplitude-distance curves on PSA surfaces show that there is a small decrease in tack and an increase in stiffness after ageing for 13 months.
The film formation of acrylic latex dispersions, containing different amounts of carboxylic acid functional groups by the incorporation of methacrylic acid (MAA), was studied with GARField 1H NMR at various relative humidities (RH). Polymer particles with glass transition temperatures in the range from 26 to 50 °C formed films at room temperature, because of hydroplasticization. It was found that with increased drying rate due to lower RH, the evaporation flux of water was limited by the latex polymer. Only in the second stage of drying this phenomenon was more obvious with increasing MAA content. 1H NMR relaxometry was used to study the change of hydrogen mobilities during film formation and hardening of the films. This showed that the drying rate itself had no impact on the hydrogen mobility in the latex films as measured via the T2 relaxation time. Hydrogen mobilities of water and the mobile polymer phase only significantly decrease after most water has evaporated. This implies that the rigidity of the polymers increases with the evaporation of water that otherwise plasticizes the polymer through hydrogen bonding with the carboxylic acid groups. This hardening of the polymer phase is essential for applications in a coating. The hydrogen mobilities were affected by the MAA concentration. Densities of mobile hydrogens increase with increasing MAA content. This is expected if the mobile protons are contained in the MAA groups. The result thus confirms the role of carboxylic acid groups in hydrogen bonding and plasticization of the copolymers. Hydrogen mobilities, however, decrease with increasing MAA content, which is hypothesized to be caused by the formation of dimers of carboxylic acid groups that still hold water. They still enable short range polymer hydrogen mobility due to hydroplasticization, but limit the long range polymer mobility due to interaction between the carboxylic acid groups.
In films cast from a colloidal dispersion comprising two particle sizes, we experimentally examine the distribution of particles normal to the substrate. The particle concentrations at various positions in the film are determined through atomic force microscopy and NMR profiling. The results are compared to a previously derived diffusional model. Evidence for diffusional driven stratification is found, but the importance of other flows is also highlighted. The conditions that enhance particle stratification are found to be a colloidally stable dispersion, low initial volume fractions, a low concentration of the stratifying particle, and for the Peclet numbers of the two components to straddle unity.
Stratified coatings are used to provide properties at a surface, such as hardness or refractive index, which are different from underlying layers. Although time-savings are offered by self-assembly approaches, there have been no methods yet reported to offer stratification on demand. Here, we demonstrate a strategy to create self-assembled stratified coatings, which can be switched to homogenous structures when required. We use blends of large and small colloidal polymer particle dispersions in water that self-assemble during drying because of an osmotic pressure gradient that leads to a downward velocity of larger particles. Our confocal fluorescent microscopy images reveal a distinct surface layer created by the small particles. When the pH of the initial dispersion is raised, the hydrophilic shells of the small particles swell substantially, and the stratification is switched off. Brownian dynamics simulations explain the suppression of stratifi-cation when the small particles are swollen as a result of reduced particle mobility, a drop in the pressure gradient, and less time available before particle jamming. Our strategy paves the way for applications in antireflection films and pro-tective coatings in which the required surface composition can be achieved on demand, simply by adjusting the pH prior to deposition.
A biocoating confines non-growing, metabolically-active bacteria within a synthetic colloidal polymer (i.e. latex) film. Bacteria encapsulated inside biocoatings can perform useful functions, such as a biocatalyst in wastewater treatment. A biocoating needs to have high a permeability to allow a high rate of mass transfer for rehydration and the transport of both nutrients and metabolic products. It therefore requires an interconnected porous structure. Tuning the porosity architecture is a challenge. Here, we exploited rigid tubular nanoclays (halloysite) and non-toxic latex particles (with a relatively high glass transition temperature) as the colloidal “building blocks” to tailor the porosity inside biocoatings containing Escherichia coli bacteria as a model organism. Electron microscope images revealed inefficient packing of the rigid nanotubes and proved the existence of nanovoids along the halloysite/polymer interfaces. Single-cell observations using confocal laser scanning microscopy provided evidence for metabolic activity of the E. coli within the biocoatings through the expression of yellow fluorescent protein. A custom-built apparatus was used to measure the permeability of a fluorescein sodium salt in the biocoatings. Whereas there was no measurable permeability in a coating made from only latex particles, the permeability coefficient of the composite biocoatings increased with increasing halloysite content up to a value of 110-4 m h-1. The effects of this increase in permeability was demonstrated through a specially-developed resazurin reduction assay. Bacteria encapsulated in halloysite composite biocoatings had statistically significant higher metabolic activities in comparison to bacteria encapsulated in a non-optimized coating made from latex particles alone.
The presence of low molar mass surfactants in latex films results in detrimental effects on their water permeability, gloss and adhesion. For applications as coatings, there is a need to develop formulations that do not contain surfactants and that have better water barrier properties. Having previously reported the synthesis of surfactant-free latex particles in water using low amounts (< 2 wt%) of controlled radical polymer chains (Lesage de la Haye et al. Macromolecules 2017, 50, 9315−9328), here we study the water barrier properties of films made from these particles and their application in anti-corrosion coatings. When films cast from aqueous dispersions of acrylate copolymer particles stabilized with poly(sodium 4-styrenesulfonate) (PSSNa) were immersed in water for three days, they sorbed only 4 wt.% water. This uptake is only slightly higher than the value predicted for the pure copolymer, indicating that the negative effects of any particle boundaries and hydrophilic stabilizing molecules are minimal. This sorption of liquid water is five times lower than what is found in films cast from particles stabilized with the same proportion of poly(methacrylic acid) (PMAA), which is more hydrophilic than PSSNa. In water vapor with 90% relative humidity, the PSSNa-based film had an equilibrium sorption of only 4 wt.%. A small increase in the PMAA content has a strong and negative impact on the barrier properties. Nuclear magnetic resonance relaxometry on polymer films after immersion in water shows that water clusters have the smallest size in the films containing PSSNa. Furthermore, these films retain their optical clarity during immersion in liquid water for up to 90 minutes, whereas all other compositions quickly develop opacity (“water whitening”) as a result of light scattering from sorbed water. This implies a remarkably complete coalescence and a very small density of defects, which yields properties matching those of some solvent borne films. The latex stabilized with PSSNa is implemented as the binder in a paint formulation for application as an anti-corrosive barrier coating on steel substrates and evaluated in accelerated weathering and corrosion tests. Our results demonstrate the potential of self-stabilized latex particles for the development of different applications, such as waterborne protective coatings and pressure-sensitive adhesives.
We advance the quantitative study of nucleation by combining the study of hundreds of samples, with statistical tests for reproducibility, and for well-defined nucleation rates. Unlike in most previous work, we find that in the samples that nucleate after the first hour of our experiment the equilibrium glycine polymorph predominates over the alpha form.
Achieving adhesion to materials with a low surface energy is a challenge when designing new soft adhesives.With this question in mind, waterborne poly(urethane-urea) (WPUU) dispersions based on poly(ethylene oxide)(PEO) homopolymer and poly(ethylene oxide-b-propylene oxide-b-ethylene oxide) (PEO-b-PPO-b-PEO) and poly(propylene oxide-b-ethylene oxide-b-propylene oxide) (PPO-b-PEO-b-PPO) triblock copolymers were prepared. Adhesives cast from the dispersions exhibited better adhesion on a low surface energy polypropylene (PP) probe than on a high surface energy steel probe. Moreover, with the aim of improving the adhesive performance of the WPUUs for pressure sensitive adhesive (PSA) applications, bilayer systems were designed. They were prepared using two WPUUs with differing viscoelastic behavior (one more solid-like and the other one, more liquid-like)to create a gradient in the viscoelastic properties. This strategy led to adhesive materials that exhibited fibrillation during probe tack measurements using a steel probe, which is typical of PSAs. Finally, prototype adhesive tapes were prepared from one of the bilayer systems. These adhesive tapes performed in a peel test in a similar way as a domestic commercial adhesive tape, making them interesting for that potential application.
The addition of cetylpyridinium chloride (CPC) to aqueous dispersions of poly(N-isopropylacrylamide) [poly(NIPAM)] and poly(N-isopropylacrylamide-co-acrylic acid) [poly(NIPAM-co-AAc)] microgel particles leads to absorption of the CPC into the particles and to corresponding changes in their hydrodynamic diameter. With the latter set of particles there is a strong pH dependence. The dependence of both hydrodynamic diameter and electrophoretic mobility of the microgel particles on the added CPC concentration show a strong correlation with CPC uptake, as obtained from direct CPC absorption measurements. Various mechanisms for CPC absorption into the microgel particles are postulated, including electrostatic, polar, and hydrophobic interactions. A comparison has also been made between the effect of added CPC on the hydrodynamic diameter of free microgel particles in dispersion, determined by dynamic light scattering, and the thickness of adsorbed monolayers of the same microgel particles deposited on cationically modified, oxidized silicon surfaces, as determined from ellipsometry measurements. The trends observed in both cases are broadly similar. This work opens the way for development of microgel layers for controlled uptake and release applications.
Templated surfaces can be used to create patterns of proteins for applications in cell biology, biosensors, and tissue engineering. A diblock copolymer template, which contains a pair of hydrophobic blocks, has been developed. The template is created from well-ordered, nonequilibrium surface structures of poly(styrene-b-isoprene) (PS-b-PI) diblock copolymers, which are achieved in ultrathin films having a thickness of less than one domain period. Adsorption and nanopatterning of bovine serum albumin (BSA) on these thin films were studied. After incubation of the copolymer templates in BSA solutions (500 μg/mL) for a period of 1 h, BSA molecules formed either a striped or a dense, ringlike structure, closely resembling the underlying polymer templates. In this “hard-soft” PS-b-PI system, BSA molecules were preferentially adsorbed on the hard PS domains, rather than on the soft PI domains. Secondary ion mass spectroscopy (SIMS) and contact angle analysis revealed that, with more PI localized at the free surface, fewer BSA molecules were adsorbed. SIMS analysis confirmed that BSA molecules were adsorbed selectively on the PS blocks. This is the first example of two hydrophobic blocks of a diblock copolymer being used as a protein patterning template. Previously reported diblock copolymer templates used hydrophilic and hydrophobic pairs. A potentially useful characteristic of this template is that it is effective at high protein solution concentrations (up to 1 mg/mL) and for long incubation times (up to 2 h), which broadens its range of applicability in various uses.
We combine experiment and theory to elucidate how small, local, structural changes can impact miscibility in polymer blends. Small-angle neutron scattering (SANS) experiments yield both the phase boundaries and the temperature dependence of the second derivative of the free energy of mixing. We demonstrate here, for the first time, that a fundamental characterization of pure component properties can be achieved through ellipsometry measurements on films of pure polymers (thickness ∼200 nm) to provide key data on the volume (or thickness)–temperature relationships; this development is significant given the scarcity of precise pressure–volume–temperature (PVT) data on pure polymers and blends. The experimental measurements allow us to undertake a detailed thermodynamic analysis of mixing using the locally correlated lattice (LCL) theory, which has been shown to be effective in rationalizing blend miscibility in terms of the pure component properties. We focus here on polymer blends of poly(α-methyl styrene-co-acrylonitrile) (PαMSAN) with deuterated poly(methyl methacrylate) (dPMMA), which differ in the degree of tacticity in the dPMMA component (atactic or syndiotactic), leading to an increase in miscibility for the latter. By combining LCL analysis of pure and mixed systems, we are able to connect tacticity changes to shifts in local nonbonded interactions, in free volume, and in thermal expansion coefficients, which in turn impact the thermodynamic compatibility of the blend components.
Hydrogen offers a source of energy that does not produce any greenhouse gas when combusted. However, some manufacturing methods of hydrogen consume large amounts of energy and produce carbon dioxide as a by-product. The production of hydrogen by bacteria is an attractive alternative, because it is not energy intensive and - under the right conditions - does not release greenhouse gases. In this review, we introduce the five known ways by which bacteria can evolve hydrogen. We then describe methods to encapsulate living bacteria in synthetic layers, called coatings, for applications in bioreactors. We review the few examples in which biocoatings have been used to produce hydrogen via the photo-fermentation method. Although not used in biocoatings so far, the dark fermentation method of hydrogen production avoids the need for illumination while offering a high yield with low oxygen evolution. We identify the potential for using genetically-modified bacteria in future research on biocoatings.
It is well known that viscosity, self-diffusion coefficient and glass transition temperature are a strong function of molecular weight (MW). Here we report the film formation characteristics of an acrylic (BMA/MMA/MAA) latex as a function of its average MW, considering four molecular weights ranging from 7,500 to 705,000 Daltons. As a means of taking into account the lower glass transition temperature and viscosity expected in the lower MW latices, we compare the film formation behaviours at the same temperature increment above their respective minimum film formation temperatures (MFT). We find that the lower MW latices form a film with a lower void concentration and lower surface roughness in comparison to the higher MW latices at the same temperature relative to the MFT. Non-invasive analysis of the film formation using ellipsometry finds evidence for void formation in the high MW latex (Mw = 705, 000 Daltons) up to 30°C above MFT. In contrast, the lowest MW latex (Mw = 7,500 Daltons) forms a dense film with few, if any, voids immediately upon the evaporation of water and at temperatures as low as 5°C above MFT. These differences can be partly attributed to varying degrees of plasticization by water. In addition, the film morphology at the polymer/air interface was investigated using atomic force microscopy. Surface features were attributed to the presence of surfactant. A low MW latex shows surfactant features at a lower film formation temperature (relative to the MFT) and/or a shorter film-formation time in comparison to the high MW latex.
ABSTRACT Biocoatings, in which viable bacteria are immobilized within a waterborne polymer coating for a wide range of potential applications, have garnered greater interest in recent years. In bioreactors, biocoatings can be ready-to-use alternatives for carbon capture or biofuel production that could be reused multiple times. Here, we have immobilized cyanobacteria in mechanically hard biocoatings, which were deposited from polymer colloids in water (i.e ., latex). The biocoatings are formed upon heating to 37°C and fully dried before rehydrating. The viability and oxygen evolution of three cyanobacterial species within the biocoatings were compared. Synechococcus sp. PCC 7002 was non-viable inside the biocoatings immediately after drying, whereas Synechocystis sp. PCC 6803 survived the coating formation, as shown by an adenosine triphosphate (ATP) assay. Synechocystis sp. PCC 6803 consumed oxygen (by cell respiration) for up to 5 days, but was unable to perform photosynthesis, as indicated by a lack of oxygen evolution. However, Chroococcidiopsis cubana PCC 7433, a strain of desiccation-resistant extremophilic cyanobacteria, survived and performed photosynthesis and carbon capture within the biocoating, with specific rates of oxygen evolution up to 0.4 g of oxygen/g of biomass per day. Continuous measurements of dissolved oxygen were carried out over a month and showed no sign of decreasing activity. Extremophilic cyanobacteria are viable in a variety of environments, making them ideal candidates for use in biocoatings and other biotechnology. IMPORTANCE As water has become a precious resource, there is a growing need for less water-intensive use of microorganisms, while avoiding desiccation stress. Mechanically robust, ready-to-use biocoatings or “living paints” (a type of artificial biofilm consisting of a synthetic matrix containing functional bacteria) represent a novel way to address these issues. Here, we describe the revolutionary, first-ever use of an extremophilic cyanobacterium ( Chroococcidiopsis cubana PCC 7433) in biocoatings, which were able to produce high levels of oxygen and carbon capture for at least 1 month despite complete desiccation and subsequent rehydration. Beyond culturing viable bacteria with reduced water resources, this pioneering use of extremophiles in biocoatings could be further developed for a variety of applications, including carbon capture, wastewater treatment and biofuel production. As water has become a precious resource, there is a growing need for less water-intensive use of microorganisms, while avoiding desiccation stress. Mechanically robust, ready-to-use biocoatings or “living paints” (a type of artificial biofilm consisting of a synthetic matrix containing functional bacteria) represent a novel way to address these issues. Here, we describe the revolutionary, first-ever use of an extremophilic cyanobacterium ( Chroococcidiopsis cubana PCC 7433) in biocoatings, which were able to produce high levels of oxygen and carbon capture for at least 1 month despite complete desiccation and subsequent rehydration. Beyond culturing viable bacteria with reduced water resources, this pioneering use of extremophiles in biocoatings could be further developed for a variety of applications, including carbon capture, wastewater treatment and biofuel production.
Films cast from multiphase polymer particles have the potential to combine the properties of their components synergistically. The properties of the film depend on the hybrid polymer architecture and the film morphology. However, how the polymer microstructure and particle morphology are transformed during film formation to determine the film morphology is not well understood. Here, using waterborne alkyd-acrylic nanocomposite particles in a case study, it was found that phase migration leading to the formation of aggregates occurred during film formation. A coarse-grained Monte Carlo model was developed to account for the effects of polymer microstructure and particle morphology on the morphology of the film. The model was validated by comparing its predictions with the observed effects, and then used to explore combinations of polymer microstructure and particle morphology not attainable with the system used as a case study. Significantly, the compatibility of the phases was found to have a greater influence than the morphology of the particles in determining the film structure.
It is shown that a combination of pulsed-field-gradient spin-echo ~PGSE! nuclear-magnetic-resonance~NMR! restricted diffusion analysis and NMR imaging may be used to measure the spatial dependence of the droplet size distribution in the cream layer of turbid oil-in-water emulsions. 1H-13C cyclic J cross-polarization PGSE is introduced as a technique for this purpose in cases where selective observation of the oil component ~or other carbohydrate constituent! is required. With this method, 13C nuclei are chemical shift selectively excited by cross-polarization from coupled 1H partners. An optimum detection sensitivity is ensured by transferring the polarization back to the coupled protons with which the combined imaging and diffusion experiment is then carried out. The spatial dependence of the oil droplet size distribution was measured for a series of emulsions containing various fractions of gum xanthan thickener dissolved in the water. The experimental results are compared with a recent model of the creaming process due to Pinfield, Dickinson, and Povey @J. Colloid Interface Sci. 166, 363 ~1994!#. When no gum xanthan is present, the experimental results are in good agreement with the model. However, the model fails to describe the droplet distribution for emulsions with a gum xanthan concentration of the order of 0.1 wt %. The discrepancy is discussed in terms of depletion flocculation and depletion stabilization.
Neutron reflection (NR), spectroscopic ellipsometry (SE), and atomic force microscopy (AFM) have been used to characterize the structure of self-assembled octadecyltrichlorosilane (OTS) layers on silicon. The first two of these techniques rely on modeling of the experimental data and may thus result in the unrealistic representation of the composition and structure at the interface. Ambiguities arise from model-dependent analysis complicated by the lack of sufficient external constraints to converge nonunique solutions to a unique one. We show in this work that AFM measurements provide extra constraints to allow us to obtain a physical description closer to the actual structure of the film. It was found that “the simpler the better” modeling strategy very often employed during the fitting of ellipsometric and neutron reflection data is, therefore, not necessarily the best way to obtain a reliable description of the interfacial structure. Our AFM findings necessitated the refit of both neutron and ellipsometric data that were previously described by a single-layer model. Interpretation of the structure of thin layers that is based only on indirect measurements such as SE, NR, and x-ray reflection techniques may be, therefore, misleading. A combined analysis of SE, NR, and AFM data suggests that the OTS film may comprise a rough layer, with pinholes down to bare silicon oxide surface, consisting at least of mono-, bi- and trilayers of OTS molecules.
Highly-mismatched III-V semiconductor alloys containing dilute concentrations of bismuth (Bi) have attracted significant attention in recent years since their unique electronic properties open up a range of possibilities for practical applications in semiconductor lasers, photovoltaics, spintronics, photodiodes, and thermoelectrics. Research on dilute bismide alloys has primarily focused to date on \text{GaAs}_{1-x}\text{Bi}_{x} , where incorporation of Bi brings about a strong reduction of the direct \Gamma -point band gap ( E_{g}{}^{\Gamma} ) -by up to 90 meV per % Bi at low Bi compositions x -characterised by strong, composition-dependent bowing. This unusual behaviour derives from the large differences in size (covalent radius) and chemical properties (electronegativity) between As and Bi.Bi, being significantly larger and more electropositive than As, acts as an isovalent impurity which primarily impacts and strongly perturbs the valence band (VB) structure. This is in contrast to dilute nitride alloys, in which small electronegative nitrogen (N) atoms strongly perturb the conduction band (CB) structure in \text{GaN}_{x}\text{As}_{1-x} and related alloys. Additionally, Bi, being the largest stable group-V element, has strong relativistic (spin-orbit coupling) effects. As such, the reduction of E_{g}{}^{\Gamma} in (\text{In})\text{GaAs}_{1-x}\text{Bi}_{x} is accompanied by a strong increase in the VB spin-orbit splitting energy ( \Delta_{\text{SO}} ).
A variety of two-dimensional (2D) nanomaterials, including graphene oxide and clays, are known to stabilize Pickering emulsions to fabricate structures for functions in sensors, catalysts, and encapsulation. We introduce here a novel Pickering emulsion using self-assembled amphiphilic triblock oligoglycine as the emulsifier. Peptide amphiphiles are more responsive to environmental changes (e.g., pH, temperature, and ionic strength) than inorganic 2D materials, which have a chemically rigid, in-plane structure. Noncovalent forces between the peptide molecules change with the environment, thereby imparting responsiveness. We provide new evidence that the biantennary oligoglycine, Gly4–NH–C10H20–NH–Gly4, self-assembles into 2D platelet structures, denoted as tectomers, in solution at a neutral buffered pH using small-angle X-ray scattering and molecular dynamics simulations. The molecules are stacked in the platelets with a linear conformation, rather than in a U-shape. We discovered that the lamellar oligoglycine platelets adsorbed at an oil/water interface and stabilized oil-in-water emulsions. This is the first report of 2D oligoglycine platelets being used as a Pickering stabilizer. The emulsions showed a strong pH response in an acidic environment. Thus, upon reducing the pH, the protonation of the terminal amino groups of the oligoglycine induced disassembly of the lamellar structure due to repulsive electrostatic forces, leading to emulsion destabilization. To demonstrate the application of the material, we show that a model active ingredient, β-carotene, in the oil is released upon decreasing the pH. Interestingly, in pH 9 buffer, the morphology of the oil droplets evolved over time, as the oligoglycine stabilizer created progressively a thicker interfacial layer. This demonstration opens a new route to use self-assembled synthetic peptide amphiphiles to stabilize Pickering emulsions, which can be significant for biomedical and pharmaceutical applications.
Postpolymerization modification is a powerful strategy to change the chemical functionality of pre-made polymers, but only limited approaches exist to modify functionality as well as the shape and behaviour of nano-particles. Herein, poly[poly(ethylene glycol) methyl ether methacrylate]-poly(2,3,4,5,6-pentafluorobenzyl methacrylate) nano-objects (pPEGMA-pPFBMA) prepared via RAFT dispersion polymerization with concurrent polymerization-induced self-assembly (PISA) in ethanol with either spherical or worm-shaped morphology were modified, post-synthesis, with a selection of 15 different thiols through thiol–para-fluoro substitution reactions in the nano-object cores. Depending on the choice of thiol, spherical nano-objects underwent an order–disorder transition to form unimers, increased in size, or underwent an order–order transition to form worm-shaped nano-objects. The core solvophobicity was found to be more important in driving a morphological transition than the modification efficiency, mass increase of the core block, or the glass transition temperature of the (partially) modified cores. These findings are relevant to the development of a “universal nanoparticle precursor” approach that allows the tuning of functionality, behaviour, size, and shape of a pre-made nano-object sample on demand.
The distribution of hydrophilic species, such as surfactants, in latex films is of critical importance for the performance of adhesives, coatings, and inks, among others. However, the evolution of this distribution during the film formation process and in the resulting dried films remains insufficiently elucidated. Here, we present in situ (wet) and ex situ (dry) small-angle neutron scattering (SANS) experiments that follow the film formation of two types of latex particles, which differ in their stabilizer: either a covalently bonded poly(methacrylic acid) (PMAA) segment or a physically adsorbed surfactant (sodium dodecyl sulfate, SDS). By fitting the experimental SANS data and combining with gravimetry experiments, we have ascertained the hydrophilic species distribution within the drying film and followed its evolution by correlating the size and shape of stabilizer clusters with the drying time. The evolution of the SDS distribution over drying time is being driven by a reduction in the interfacial free energy. However, the PMAA-based stabilizer macromolecules are restricted by their covalent bonding to core polymer chains and hence form high-surface area disclike phases at the common boundary between particles and PMAA micelles. Contrary to an idealized view of film formation, PMAA does not remain in the walls of a continuous honeycomb structure. The results presented here shed new light on the nanoscale distribution of hydrophilic species in drying and ageing latex films. We provide valuable insights into the influence of the stabilizer mobility on the final structure of latex films.
The adhesion of Staphylococcus epidermidis, Escherichia coli, and Candida albicans on mucin coatings was evaluated to explore the feasibility of using the coating to increase the infection resistance of biomaterials. Coatings of bovine submaxillary mucin (BSM) were deposited on a base layer consisting of a poly(acrylic acid-b-methyl methacrylate) (PAA-b-PMMA) diblock copolymer. This bi-layer system exploits the mucoadhesive interactions of the PAA block to aid the adhesion of mucin to the substratum, whereas the PMMA block prevents dissolution of the coating in aqueous environments. The thickness of the mucin coating was adjusted by varying the pH of the solution from which it was deposited. Thin mucin coatings decreased the numbers of bacteria but increased the numbers of C. albicans adhering to the copolymer and control surfaces. Increasing the mucin film thickness resulted in a further lowering of the density of adhering S. epidermidis cells, but it did not affect the density of E. coli. In contrast, the density of C. albicans increased with an increase in mucin thickness.
Oil-in-water emulsions, stabilised with conventional surfactants, are commonly used in eye drops for ocular drug delivery. However, the presence of surfactants can sometimes irritate tissues. Furthermore, conventional emulsions often have poor retention on ocular tissue. Pickering emulsions stabilised with nanoparticles have been gaining attention in recent years for a range of biomedical applications because of their biocompatibility. Here, Pickering emulsions were evaluated for the first time for the confinement of organic components for potential application in ocular drug delivery. For a model system, we used nanodiamond (ND) nanoparticles functionalised with covalently-bonded two-tail (2T) oligoglycine C-10(NGly(4))(2) to make Pickering oil-in-water emulsions, which were stable over three months of storage under neutral pH. We proved the non-toxicity of ND-2T Pickering emulsions, comparable to buffer solution, via an ex vivo bovine corneal permeability and opacity test. The retention of the oil phase in the ND-2T stabilised emulsions on corneal tissue is significantly increased because of the mucoadhesive properties arising from the positively-charged terminal amino groups of 2T. Our formulated emulsions have a surface tension, pH and salt concentration comparable to that of tear fluid. The high retention of the ND-2T-stabilised emulsions on the corneal surface, in combination with their non-toxicity, gives them distinct advantages for ocular drug delivery. The principles of this model system could be applied in the future design of a range of formulations for drug delivery.
This article provides an overview of the outcomes of a European-funded project called BarrierPlus. A new type of water-based barrier coating was developed for structural steel applications. The advantages of this coating include enhanced moisture resistance, low volatile organic compounds (VOCs) and one-component self-crosslinking free of isocyanates. To enable this performance, a latex polymer binder was uniquely designed without using soap-like molecules, known as surfactants, to form the dispersion. By minimizing surfactants in the coating, the barrier properties were significantly enhanced. The latex was successfully scaled up to 15 kg quantities by an SME, coating formulations were scaled to pilot quantities and a variety of characterization and coating performance tests were completed. A life cycle assessment found that the BarrierPlus coating has a better environmental profile than an industry benchmark solvent-borne coating and showed promising results relative to commercial waterborne benchmarks.
A major drawback of conventional emulsion polymers arises from the presence of migrating low molecular weight surfactants that contribute to poor water barrier properties and low adhesion to substrates. In this paper, we demonstrate how living polymer chains obtained by reversible addition-fragmentation chain transfer (RAFT) can be used as an efficient stabilizer in emulsion polymerization, leading to the production of surfactant-free latexes, which then form crosslinked films with beneficial properties. Hydrophilic poly(methacrylic acid) (PMAA) chains obtained by RAFT performed in water are used to mediate emulsion polymerization and produce film-forming latex particles from mixtures of methyl 2 methacrylate, n-butyl acrylate and styrene. Stable dispersions of particles with sizes between 100 and 200 nm are obtained, with very low amounts of coagulum (< 0.5 wt.%). The particles are stabilized by the PMAA segment of amphiphilic block copolymers formed during the polymerization. Remarkably, low amounts of PMAA chains (from 1.5 wt.% down to 0.75 wt.%) are enough to ensure particle stabilization. Only traces of residual PMAA macroRAFT agents are detected in the final latexes, showing that most of them are successfully chain extended and anchored on the particle surface. The Tg of the final material is adjusted by the composition of the hydrophobic monomer mixture so that film formation occurs at room temperature. Conventional crosslinking strategies using additional hydrophobic co-monomers, such as 1,3-butanediol diacrylate (BuDA), diacetone acrylamide (DAAm), and (2-acetoacetoxy)ethyl methacrylate, are successfully applied to these formulations as attested by gel fractions of 100%. When particles are internally crosslinked with BuDA, chain interdiffusion between particles is restricted, and a weak and brittle film is formed. In contrast, when DAAm undergoes crosslinking during film formation, full coalescence is achieved along with the creation of a crosslinked network. The resulting film has a higher Young’s modulus and tensile strength as a result of crosslinking. This synthetic strategy advantageously yields a surfactant-free latex that can be formed into a film at room temperature with mechanical properties that can be tuned via the crosslinking density.
The isothermal structural relaxation of glassy, spin-cast polymer thin films has been investigated. Specifically, the thickness, h, of freshly-cast poly(methyl methacrylate) thin films was measured over time using spectroscopic ellipsometry. The spin-cast films exhibit a gradual decrease in thickness, which is attributed to structural relaxation of the glass combined with simultaneous solvent loss. In all cases, h was found to be greater than the equilibrium thickness, h0, which is obtained by cooling slowly from the melt. It is observed that both the rate of the volume relaxation and the fractional departure from h0 (referred to as ∂o) increase with increasing film thickness. In the limit of very thin films, the initial h is close to h, and o is small, whereas in thick films (> 500 nm), a plateau value of ∂o of 0.16 is observed, which is close to the volume fraction of the solvent at the vitrification point. This dependence of ∂o on thickness is observed regardless of the substrate, polymer molecular weight, or angular velocity during spin-casting. Enhanced mobility near film surfaces could be leading to greater relaxation in thinner films prior to, and immediately after, the vitrification of the polymer during the deposition process.
Polymer coatings with periodic topographic patterns, repeating over millimetre length scales, can be created from lateral flows in an aqueous dispersion of colloidal particles. The flow is driven by differences in evaporation rate across the wet film surface created by IR radiative heating through a shadow mask. This new process, which we call IR radiation-assisted evaporative lithography (IRAEL), combines IR particle sintering with the concept of evaporative lithography. Here, a series of experiments has been conducted in which the mass of the latex is measured as a function of the exposure time under infrared radiation through a mask. The water evaporation rates and the minimum exposure time required for a dry film are estimated as a function of the power density of the IR emitter. The temperature of the wet film is monitored to avoid overheating and boiling of the water, which will otherwise cause defects. It is demonstrated that textured films can be created on a variety of substrates (plastics, metals, paper and glass), and processing times can be as short as 5 min. We use IRAEL to decorate household goods with an aesthetic coating with the desired texture. © 2013 Elsevier B.V. All rights reserved.
We present a simple technique to switch off the tack adhesion in selected areas of a colloidal nanocomposite adhesive. It is made from a blend of soft colloidal polymer particles and hard copolymer nanoparticles. In regions that are exposed to IR radiation, the nanoparticles sinter together to form a percolating skeleton, which hardens and stiffens the adhesive. The tack adhesion is lost locally. Masks can be made from silicone-coated disks, such as coins. Under the masks, adhesive island regions are defined with the surrounding regions being a nontacky coating. When optimizing the nanocomposite's adhesive properties, the addition of the hard nanoparticles raises the elastic modulus of the adhesive significantly, but adhesion is not lost because the yield point remains relatively low. During probe-tack testing, the soft polymer phases yield and enable fibrillation. After heating under IR radiation, the storage modulus increases by a factor of 5, and the yield point increases nearly by a factor of 6, such that yielding and fibrillation do not occur in the probe-tack testing. Hence, the adhesion is lost. Loading and unloading experiments indicate that a rigid skeleton is created when the nanoparticles sinter together, and it fractures under moderate strains. This patterning method is relatively simple and fast to execute. It is widely applicable to other blends of thermoplastic hard nanoparticles and larger soft particles.
This book introduces the reader to latex, which is a colloidal dispersion of polymer particles in water, and explains how useful products are made from it.
The coating of substrates with an extracellular matrix (ECM) protein, such as fibronectin (FN), is often employed to increase cell adhesion and growth. Here, we examine the influence of the size scale and geometry of novel FN nanopatterns on the adhesion and spreading of Chinese Hamster Ovary (CHO) cells. The FN is patterned on the surface of templates created through the self-assembly of polystyrene-block-polyisoprene (PS-b-PI) diblock copolymers. Both ring-like and stripe-like FN nanopatterns are created through the preferential adsorption of FN on PS blocks, as confirmed through the complementary use of atomic force microscopy and secondary ion mass spectrometry. The ring-like FN nanopattern substrate increases the cells' adhesion compared with the cells on homogeneous FN surfaces and the stripe-like FN nanopatterns. Cell adhesion is high when the FN ring size is greater than 50 nm and when the surface coverage of FN is less than ca. 85%. We suggest that the ring-like nanopatterns of FN may be aiding cell adhesion by increasing the clustering of the proteins (integrins) with which cells bind to the nanopatterned substrate. This clustering is required for cell adhesion. In comparison to lithographic techniques, the FN templating method, presented here, provides a simple, convenient and economical way of coating substrates for tissue cultures and should be applicable to tissue engineering.
Developing the use of polymers from renewable sources to build hydrogels with tailored mechanical properties has become an increasing focus of research. The impact of the thermo-reversible physical networks of gelatin (arising from the formation of triple-helices) on the structure formation of a chemical network, obtained by cross-linking with glutaraldehyde (a non-catalytic cross-linker), was studied using optical rotation, oscillatory rheology and large strain mechanical deformation. We observed a direct correlation between the storage shear modulus of the chemical network grown in the gel state (i.e. simultaneously with the physical network) and the amount of gelatin residues in the triple-helix conformation (χ). Since χ is directly affected by temperature, the value of the storage modulus is also sensitive to changes in the temperature of gel formation. χ values as low as 12% lead to an increase of the shear storage modulus of the cross-linked gel by a factor of 2.7, when compared to a chemical network obtained in the sol state (i.e. in the absence of a physical network). Our results show that the physical network acts as a template, which leads to a greater density of the chemical cross-links and a corresponding higher elastic modulus, beyond what is otherwise achieved in the absence of a physical network.
Poly(chloroprene) is a synthetic crystallizable polymer used in several applications, including rubber gloves. The film formation of poly(chloroprene) latex offers opportunities to define structures at length scales between the molecular and macroscopic, thereby adjusting the elastomer’s mechanical properties. However, the connections between processing and the resultant film properties are not fully understood. Here, we investigate the competition between the coalescence of latex particles to build cohesive strength and their crystallization to raise the elastic modulus. We demonstrate that when coalescence precedes crystallization, the elastomer has greater extensibility and a higher tensile strength compared to when crystallization occurs during coalescence. The mechanical properties of poly(chloroprene) were tuned by blending two colloids with differing gel contents and crystallizabilities. Heating above poly(chloroprene)’s melting temperature allows increased particle interdiffusion and builds cohesion, prior to recrystallization. We provide evidence from in situ wide-angle X-ray scattering for the strain-induced crystallization of as-cast films from particle blends.
Strategies for synthesizing molecularly designed materials are expanding, but methods for their thermodynamic characterization are not. This shortfall presents a challenge to the goal of connecting local molecular structure with material properties and response. Fundamental thermodynamic quantities, including the thermal expansion coefficient, α, can serve as powerful inputs to models, yielding insight and predictive power for phenomena ranging from miscibility to dynamic relaxation. However, the usual routes for thermodynamic characterization often require a significant sample size (e.g. one gram), or challenging experimental set-ups (e.g. mercury as a confining fluid), or both. Here, we apply spectroscopic ellipsometry, which is an optical technique for thin film analysis, to obtain thermodynamic data. We clarify issues in the scientific literature concerning the connection between ellipsometric and volumetric thermal expansion coefficients for substances in both the glass and melt states. We analyze temperature-dependent data derived using both ellipsometry and macro-scale dilatometric techniques for ten different polymers. We find superb correlation between the α values obtained via the two techniques, after considering the effects of mechanical confinement by the substrate for a glassy thin film. We show how the ellipsometric α can serve as input to the locally correlated lattice theory to yield predictions for the percent free volume in each polymer as a function of temperature. We find that the ellipsometric α at the glass transition temperature, Tg, is not only material dependent, but it is linearly correlated with Tg itself. Spectroscopic ellipsometry, which requires only very small quantities of sample and is straightforward to perform, will significantly expand the range of systems for which thermodynamic properties can be characterized. It will thus advance our ability to use theory and modeling to predict the miscibility and dynamic relaxation of new materials. As such, ellipsometry will be able to underpin materials synthesis and property design.
The molecular composition of polymer blend surfaces defines properties such as adhesion, wetting, gloss, and biocompatibility. The surface composition often differs from the bulk because of thermodynamic effects or modification. Mixtures of colloids and linear polymers in a common solvent are often used to deposit films for use in encapsulants, inks, coatings, and adhesives. However, means to control the nonequilibrium surface composition are lacking for these systems. Here we show how the surface composition and hydrophilicity of a film deposited from a bimodal mixture of linear polymers and colloids in water can be adjusted simply by varying the evaporation rate. Ion beam analysis was used to quantify the extent of stratification of the linear polymers near the surface, and the results are in agreement with a recent diffusiophoretic model. Because our approach to stratification relies solely on diffusiophoresis, it is widely applicable to any system deposited from colloids and nonadsorbing polymers in solution as a means to tailor surface properties.
The widespread use of thin films in a range of applications and industries, from coatings, inks and lithography to nano-imprinting, optoelectronics and memory devices,1 has made the understanding of thin films, particularly the changes induced by structural relaxation and solvent evaporation, very important. There is a need to know whether a film will change in dimensions after its deposition and how fast these changes will occur.
For a wide range of applications, films are deposited from colloidal particles suspended in a volatile liquid. There is burgeoning interest in stratifying colloidal particles into separate layers within the final dry film to impart properties at the surface different to the interior. Here, we outline the mechanisms by which colloidal mixtures can stratify during the drying process. The problem is considered here as a three-way competition between evaporation of the continuous liquid, sedimentation of particles, and their Brownian diffusion. In particle mixtures, the sedimentation of larger or denser particles offers one means of stratification. When the rate of evaporation is fast relative to diffusion, binary mixtures of large and small particles can stratify with small particles on the top, according to physical models and computer simulations. We compare experimental results found in the scientific literature to the predictions of several recent models in a quantitative way. Although there is not perfect agreement between them, some general trends emerge in the experiments, simulations and models. The stratification of small particles on the top of a film is favoured when the colloidal suspension is dilute but when both the concentration of the small particles and the solvent evaporation rate are sufficiently high. A higher ratio also favours stratification by size. This review points to ways that microstructures can be designed and controlled in colloidal materials to achieve desired properties.
In waterborne mixtures of colloidal particles with differing sizes, the spontaneous stratification of one species of particle in a coating – driven by diffusiophoresis - offers the possibility to tailor the surface properties. However, despite strong research interest in stratification in recent years, the acceptable range of experimental parameters has not been fully explored, and the extent of stratification that is achievable has not yet been quantified. Here, we study the stratification of bimodal mixtures of waterborne polyurethane particles mixed with larger acrylic particles. We use ultra-low angle microtoming to prepare cross-sections of coating samples and analyse compositions quantitatively with Raman mapping. We use this method to obtain high-resolution depth profiles of the polyurethane phase in the coating with spacing between measurements corresponding to a few tens of nm. We experimentally test a model of diffusiophoresis and observe stratification when the processing parameters (evaporation rates, film thickness, and volume fraction of small particles) fall within the required range. Samples that exhibit stratification have top layer thicknesses on the order of tens of μm, which is a significant depth for exploitation in coatings aiming to modify surface properties. To guide the design of coatings in applications, we draw on the model to define the range of parameters in which self-stratification is expected. Our results provide a fundamental understanding that will enable the fabrication of tailored coatings in which the properties of the surface differ from the bulk material.
Dip coating is a very common process in food manufacturing. Controlling the thickness of the coating is key to deliver the desired sensorial properties and to be compliant with the product’s nutritional claims. Whilst dip coating with Newtonian liquids is physically well understood, coating food products almost invariably involve liquids with more complex rheology. This makes the process more difficult to design and control and reduces the coating homogeneity. Developing novel food products with improved nutritional attributes often calls for reducing the coating thickness and non-homogeneity should be avoided to guarantee the quality of the final product. In this study, we focused on the coating of a flat surface using Carbopol solutions and a commercial ketchup, following a Herschel-Bulkley rheological model. The final average coating thickness was always significantly lower than the critical thickness that can be estimated from liquid density and yield stress. Liquids with a yield stress in the range 4-56 Pa were considered in this study and the steady withdrawal speed from the bath was varied in the range 0.1-20 mm/s. The resulting average coating thickness and its uniformity are discussed. The results are interpreted in the context of an existing theory for dip coating with liquids with a yield stress. This study paves the way toward an integrated design of the coating process and the liquid rheology of foods, such as chocolate or ketchup. This can enable the development of new food products allying improved nutrition, a consumer preferred sensory profile and cost.
Variable angle spectroscopic ellipsometry is a nondestructive technique for accurately determining the thicknesses and refractive indices of thin films. Experimentally, the ellipsometry parameters ψ and Δ are measured, and the sample structure is then determined by one of a variety of approaches, depending on the number of unknown variables. The ellipsometry parameters have been inverted analytically for only a small number of sample types. More general cases require either a model-based numerical technique or a series of approximations combined with a sound knowledge of the test sample structure. In this paper, the combinatorial optimization technique of simulated annealing is used to perform least-squares fits of ellipsometry data (both simulated and experimental) from both a single layer and a bilayer on a semi-infinite substrate using what is effectively a model-free system, in which the thickness and refractive indices of each layer are unknown. The ambiguity inherent in the best-fit solutions is then assessed using Bayesian inference. This is the only way to consistently treat experimental uncertainties along with prior knowledge. The Markov chain Monte Carlo algorithm is used. Mean values of unknown parameters and standard deviations are determined for each and every solution. Rutherford backscattering spectrometry is used to assess the accuracy of the solutions determined by these techniques. With our computer analysis of ellipsometry data, we find all possible models that adequately describe that data. We show that a bilayer consisting of a thin film of poly(styrene) on a thin film of silicon dioxide on a silicon substrate results in data that are ambiguous; there is more than one acceptable description of the sample that will result in the same experimental data.
In simulations and experiments, we study the drying of films containing mixtures of large and small colloidal particles in water. During drying, the mixture stratifies into a layer of the larger particles at the bottom with a layer of the smaller particles on top. We developed a model to show that a gradient in osmotic pressure, which develops dynamically during drying, is responsible for the segregation mechanism behind stratification.
Soft adhesives require an optimum balance of viscous and elastic properties. Adhesion is poor when the material is either too solidlike or too liquidlike. The ability to switch tack adhesion off at a desired time has many applications, such as in recycling, disassembly of electronics, and painless removal of wound dressings. Here, we describe a new strategy to switch off the tack adhesion in a model nanocomposite adhesive in which temperature is the trigger. The nanocomposite comprises hard methacrylic nanoparticles blended with a colloidal dispersion of soft copolymer particles. At relatively low volume fractions, the nanoparticles (50 nm diameter) accumulate near the film surface, where they pack around the larger soft particles (270 nm). The viscoelasticity of the nanocomposite is adjusted via the nanoparticle concentration. When the nanocomposite is heated above the glass transition temperature of the nanoparticles (T(g) = 130 °C), they sinter together to create a rigid network that raises the elastic modulus at room temperature. The tackiness is switched off. Intense infrared radiation is used to heat the nanocomposites, leading to a fast temperature rise. Tack adhesion is switched off within 30 s in optimized compositions. These one-way switchable adhesives have the potential to be patterned through localized heating.
It might reasonably be expected that semiconductor devices would inevitably displace thermal devices in all aspects of gas sensing. However, thermal sources and detectors are inexpensive and suffice for a very wide range of gas sensing applications. The advent of thin film sources provided the necessary extended wavelength coverage beyond 4.5 µm compared with glass-envelope filament lamps, and the emitted power is generally higher for thermal sources. Because of the complexity of fabricating semiconductor devices with the materials used, there is a significant economic barrier to them entering the marketplace. They do offer the advantages of being robust and having a long lifetime and, when used as both source and detector, much higher modulation frequencies are available. The temperature dependence of semiconductor devices is higher than thermal devices, which requires temperature stabilisation, or compensation. Incorporating a modest cooling with the temperature stabilisation in semiconductor devices would be advantageous but at the expense of electrical power and some complexity. It is, perhaps, where reliability is paramount that semiconductor systems are likely to be introduced.
Hypothesis Surfactants in emulsions sometimes do not provide adequate stability against coalescence, whereas Pickering emulsions often offer greater stability. In a search for stabilizers offering biocompatibility, we hypothesized that carboxylated nanodiamonds (ND) would impart stability to Pickering emulsions. Experiments We successfully prepared Pickering emulsions of sunflower oil in water via two different methods: membrane emulsification and probe sonication. The first method was only possible when the pH of the aqueous ND suspension was ≤4. Findings Pendant-drop tensiometry confirmed that carboxylated ND is adsorbed at the oil/water interface, with a greater decrease in interfacial tension found with increasing ND concentrations in the aqueous phase. The carboxylated ND become more hydrophilic with increasing pH, according to three-phase contact angle analysis, because of deprotonation of the carboxylic acid groups. Membrane emulsification yielded larger (about 30 µm) oil droplets, probe sonication produced smaller (sub-μm) oil droplets. The Pickering emulsions show high stability against mechanical vibration and long-term storage for one year. They remain stable against coalescence across a wide range of pH values. Sonicated emulsions show stability against creaming. In this first-ever systematic study of carboxylated ND-stabilized Pickering emulsions, we demonstrate a promising application in the delivery of β-carotene, as a model active ingredient.
Waterborne coatings emit a low amount of harmful volatile organic compounds (VOCs) into the atmosphere compared to solvent-cast coatings. A typical waterborne formulation for agricultural applications consists of colloidal thermoplastic particles (latex) as the binder, a thickener to raise the viscosity, inorganic filler particles with a water-soluble dispersant, and a colloidal wax to modify surface properties. The formulations typically contain hygroscopic species that are potentially subject to softening by environmental moisture. The hardness, tack adhesion, and coefficient of friction of formulated coatings determines their suitability in applications. However, the relationship of these properties to the components in a coating formulation has not been adequately explored. Furthermore, the relationship between hygroscopic components and properties is an added complication. Here, we have characterized the hardness and tack adhesion of model formulated coatings using a single micro-indentation cycle with a conical indenter under controlled temperatures (above and below the glass transition temperature of a latex binder) and relative humidities. In parallel, we measured the coefficient of kinetic friction, μk, for the same coatings using a bespoke testing rig under controlled environmental conditions. Across a range of temperatures, RH and compositions, we find an inverse correlation between the coating hardness and μk. Any correlation of μk with the roughness of the coatings, which varies with the composition, is less clear. Formulations that contain wax additives have a higher μk at a low RH of 10%, in comparison to formulations without wax. For the wax formulations, μk decreases when the RH is raised, whereas in non-wax formulations, μk increases with increasing RH. Wax-containing coatings are hydrophilic (with a lower water contact angle), however the wax has a lower water permeability. A lubricating layer of water can explain the lower observed μk in these formulations. The addition of wax is also found to planarize the coating surface, which leads to higher tack adhesion in dry coatings in comparison to coatings without wax. Greater adhesive contact in these coatings can explain their higher friction. Our systematic research will aid the design of seed coating formulations to achieve their optimum properties under a wide range of environmental conditions.
PEGylated organosilica nanoparticles have been synthesized through self-condensation of 3-mercaptopropyltrimethoxysilane in dimethylsulfoxide into thiolated nanoparticles with their subsequent reaction with methoxypolyethylene glycol maleimide. The PEGylated nanoparticles showed excellent colloidal stability over a wide range of pHs in contrast to the parent thiolated nanoparticles, which have a tendency to aggregate irreversibly under acidic conditions (pH < 3.0). Due to the presence of a poly(ethylene glycol)-based corona, the PEGylated nanoparticles are capable of forming hydrogen-bonded interpolymer complexes with poly(acrylic acid) in aqueous solutions under acidic conditions, resulting in larger aggregates. The use of hydrogen-bonding interactions allows their more efficient attachment of the nanoparticles to surfaces. The alternating deposition of PEGylated nanoparticles and poly(acrylic acid) on silicon wafer surfaces in a layer-by-layer fashion leads to multilayered coatings. The self-assembly of PEGylated nanoparticles with poly(acrylic acid) in aqueous solutions and at solid surfaces was compared to the behavior of linear poly(ethylene glycol). The nanoparticle system creates thicker layers than the poly(ethylene glycol), and a thicker layer is obtained on a poly(acrylic acid) surface than on a silica surface, because of the effects of hydrogen bonding. Some implications of these hydrogen bonding-driven interactions between PEGylated nanoparticles and poly(acrylic acid) for pharmaceutical formulations are discussed.
The length scales of film, thickness nonuniformities, commonly observed in polymer colloid (i.e., latex) films, are predicted. This prediction is achieved by investigating the stability behavior of drying latex films. A linear stability analysis is performed on a base solution representing a uniformly drying latex film containing a surfactant. The analysis identifies film thickness nonuniformities over two length scales: long (millimeter) range (from lubrication theory) and short (micrometer) range (from nonlubrication theory). Evaporation and surfactant desorption into the bulk film are identified as the primary destabilizing mechanisms during drying. Experimental evidence through direct visualization and atomic force microscopy confirm the existence of nonuniformities over both length scales, which are shown to be functions of parameters such as initial particle volume fraction, surfactant amount, and desorption strength, while being independent of drying rate. © 2008 American Institute of Chemical Engineers.
Critical point transition energies and optical functions of the novel GaAs-based dilute bismide alloys GaAsBi, GaNAsBi, and GaPAsBi were determined using spectroscopic ellipsometry. The ellipsometry data were analyzed using a parameterized semiconductor model to represent the dielectric function of the alloys as the sum of Gaussian oscillators centered on critical points in the band structure, and from this extracting the energies of those critical points. The band gap and spin-orbit splitting were measured for samples for a range of alloy compositions. The first experimental measurements of the spin-orbit splitting in the GaNAsBi quaternary alloy were obtained, which showed that it is approximately independent of N content, in agreement with theory. The real component of the refractive index in the transparent region below the band gap was found to decrease as the band gap increased for all of the alloys studied, following the usual relations for conventional semiconductors. This work provides key electronic and optical parameters for the development of photonic devices based on these novel alloys. Published by AIP Publishing.
Nanocomposites of a polymer and carbon nanotubes exhibit high electrical and thermal conductivity and enhanced mechanical properties in comparison to the polymer alone. Film formation from latex dispersions is an ideal way to create nanocomposite coatings with the advantages of solvent-free processing and a high uniformity of dispersion. It is shown here that carbon nanotubes functionalised with poly(vinyl alcohol) (PVA) can be blended with two types of acrylic latex to create stable colloidal dispersions without the need for added surfactant or emulsifier. Waterborne nanocomposite films with optical transparency can be formed. Microscopic analysis shows that the PVA-functionalized nanotubes are finely and uniformly dispersed in the polymer matrix.
Most substances can crystallise into two or more different crystal lattices, called polymorphs. Despite this, there are no systems in which we can quantitatively predict the probability of one competing polymorph forming, instead of the other. We address this problem using large scale (hundreds of events) studies of the competing nucleation of the alpha and gamma polymorphs of glycine. In situ Raman spectroscopy is used to identify the polymorph of each crystal. We find that the nucleation kinetics of the two polymorphs is very different. Nucleation of the alpha polymorph starts off slowly but accelerates, while nucleation of the gamma polymorph starts off fast but then slows. We exploit this difference to increase the purity with which we obtain the gamma polymorph by a factor of ten. The statistics of the nucleation of crystals is analogous to that human mortality, and using a result from medical statistics we show that conventional nucleation data can say nothing about what, if any, are the correlations between competing nucleation processes. Thus we can show that it is impossible to disentangle the competing nucleation processes. We also find that the growth rate and the shape of a crystal depends on when it nucleated. This is new evidence that nucleation and growth are linked.
Both visible and infrared (IR) spectroscopic ellipsometry have been employed to study the structure of thin layers of bovine submaxillary mucin (BSM) adsorbed on poly(acrylic acid-block-methyl methacrylate) (PAA-b-PMMA) copolymer and poly(methyl methacrylate) (PMMA) surfaces at three pH values (3, 7, and 10). The adsorbed mucin layer on the copolymer surface had the greatest thickness (17 nm) when adsorbed from a mucin solution at a pH of 3. For the first time, IR ellipsometry was used to identify adhesive interactions and conformational changes in mucin/polymer double layers. After applying the regularized method of deconvolution in the analysis, the formation of hydrogen bonds between the carboxyl groups of the BSM and PAA-b-PMMA copolymer in double layers has been found. The IR ellipsometry data, in agreement with the visible ellipsometry analysis, indicate the pH dependence of adhesion of mucin to the copolymer surface. There is an increase in the amount of hydrogen-bonded carboxyl groups in mucin deposited at a pH of 3. There is no evidence that the amide groups of the mucin participate in this bonding. At the lower pH, the IR ellipsometry spectra after deconvolution reveal an increase in the proportion of β-sheets in the BSM upon adsorption on the copolymer surface, indicating a more unfolded, aggregated structure. The IR ellipsometry data also indicated some changes in the conformational states of the side groups in the copolymer induced by entanglements and bonding interactions with the mucin macromolecules. Deconvolution provides an unprecedented level of information from the IR ellipsometry spectra and yields important insights. © 2009 Society for Applied Spectroscopy.
The use of 2,3,4,5,6-pentafluorobenzyl methacrylate (PFBMA) as a core-forming monomer in ethanolic RAFT dispersion polymerization formulations is presented. Poly[poly(ethylene glycol) methyl ether methacrylate] (pPEGMA) macromolecular chain transfer agents were chain extended with PFBMA leading to nanoparticle formation via polymerization-induced self-assembly (PISA). pPEGMA-pPFBMA particles exhibited the full range of morphologies (spheres, worms, and vesicles) including pure and mixed phases. Worm phases formed gels that underwent a thermo-reversible degelation and morphological transition to spheres (or spheres and vesicles) upon heating. Post-synthesis, the pPFBMA cores were modified through thiol–para-fluoro substitution reactions in ethanol using 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) as the base. For monothiols, conversions were 64% (1-octanethiol) and 94% (benzyl mercaptan). Spherical and worm-shaped nano-objects were core cross-linked using 1,8-octanedithiol, which prevented their dissociation in non-selective solvents. For a temperature-responsive worm sample, cross-linking additionally resulted in the loss of the temperature-triggered morphological transition. The use of the reactive monomer PFBMA in PISA formulations presents a simple method to prepare well-defined nano-objects similar to those produced with non-reactive monomers (e.g. benzyl methacrylate) and to retain morphologies independent of solvent and temperature.
Using spectroscopic ellipsometry measurements on GaP1−χBiχ/GaP epitaxial layers up to χ = 3.7% we observe a giant bowing of the direct band gap (EΓg) and valence band spin-orbit splitting energy (ΔSO). EΓg (ΔSO) is measured to decrease (increase) by approximately 200 meV (240 meV) with the incorporation of 1% Bi, corresponding to a greater than fourfold increase in ΔSO in going from GaP to GaP0.99Bi0.01. The evolution of EΓg and ΔSO with χ is characterised by strong, composition-dependent bowing. We demonstrate that a simple valence band-anticrossing model, parametrised directly from atomistic supercell calculations, quantitatively describes the measured evolution of EΓg and ΔSO with χ. In contrast to the well-studied GaAs1−χBiχ alloy, in GaP1−χBiχ substitutional Bi creates localised impurity states lying energetically within the GaP host matrix band gap. This leads to the emergence of an optically active band of Bi-hybridised states, accounting for the overall large bowing of EΓg and ΔSO and in particular for the giant bowing observed for χ ≲ 1%. Our analysis provides insight into the action of Bi as an isovalent impurity, and constitutes the first detailed experimental and theoretical analysis of the GaP1−χBiχ alloy band structure.
We report the first demonstration of a solid-state, direct-conversion sensor for thermal neutrons based on a polymer/inorganic nanocomposite. Sensors were fabricated from ultra-thick films of poly(triarylamine) (PTAA) semiconducting polymer, with thicknesses up to 100 μm. Boron nanoparticles were dispersed throughout the PTAA film to provide the neutron stopping power arising from the high thermal neutron cross-section of the isotope Boron-10. To maximize the quantum efficiency of the sensor to thermal neutrons, a high volume fraction of homogeneously dispersed boron nanoparticles was achieved in the thick PTAA film using an optimized processing method. Thick active layers were realized using a high molecular weight of the PTAA so that molecular entanglements provide a high cohesive strength. A non-ionic surfactant was used to stabilize the boron dispersion in solvent and hence suppress the formation of agglomerates and associated electrical pathways. Boron nanoparticle loadings of up to ten volume percent were achieved, with thermal neutron quantum efficiency estimates up to 6% resulting. The sensors' neutron responses were characterized under a high flux thermal neutron exposure, showing a linear correlation between the response current and the thermal neutron flux. Polymer-based boron nanocomposite sensors offer a new neutron detection technology that uses low-cost, scalable solution processing, and provides an alternative to traditional neutron sensors that use rare isotopes, such as Helium-3.
Semiconducting polymer X-radiation detectors are a completely new family of low-cost radiation detectors with potential application as beam monitors or dosimeters. These detectors are easy to process, mechanically flexible, relatively inexpensive, and able to cover large areas. However, their x-ray photocurrents are typically low as, being composed of elements of low atomic number (Z), they attenuate x-rays weakly. Here, the addition of high-Z nanoparticles is used to increase the x-ray attenuation without sacrificing the attractive properties of the host polymer. Two types of nanoparticles (NPs) are compared: metallic tantalum and electrically insulating bismuth oxide. The detection sensitivity of 5 µm thick semiconducting poly([9,9-dioctylfluorenyl-2,7-diyl]-co-bithiophene) diodes containing tantalum NPs is four times greater than that for the analogous NP-free devices; it is approximately double that of diodes containing an equal volume of bismuth oxide NPs. The x-ray induced photocurrent output of the diodes increases with an increased concentration of NPs. However, contrary to the results of theoretical x-ray attenuation calculations, the experimental current output is higher for the lower-Z tantalum diodes than the bismuth oxide diodes, at the same concentration of NP loading. This result is likely due to the higher tantalum NP electrical conductivity, which increases charge transport through the semiconducting polymer, leading to increased diode conductivity.
Hypothesis Colloidal polymer composites, in which polymer particles are blended with a filler, are widely used in applications including pharmaceuticals, crop protection, inks, and protective coatings. It is generally found that the presence of hard particulate fillers will increase the elastic modulus of a polymer colloid composite. However, the influence of the size of the filler particle on the large-strain deformation and fracture and on the viscoelastic characteristics, including creep, is not well explored. We hypothesize that the size ratio of the filler to the colloidal polymer will play a critical role in determining the properties of the composite. Experiments Colloidal composites were prepared by blending soft polymer colloids (as a binder) with calcium carbonate fillers having four different sizes, spanning from 70 nm to 4.5 m. There is no bonding between the filler and matrix in the composites. The large-strain deformation, linear viscoelasticity, and creep were determined for each filler size for increasing the filler volume fractions (CC). Weibull statistics were used to analyze the distributions of strains at failure. Findings We find that the inclusion of nano-fillers leads to brittle fracture at a lower CC than when m-size fillers are used. The data interpretation is supported by Weibull analysis. However, for a given CC, the storage modulus is higher in the rubbery regime, and the creep resistance is higher when nanoparticles are used. Using scanning electron microscopy to support our arguments, we show that the properties of colloidal composites are correlated with their microstructure, which can be altered through control of the filler:polymer particle size ratio. Hard nanoparticles pack efficiently around larger particles to provide reinforcement (manifested as a higher storage modulus and greater creep resistance), but they also introduce weak points that lead to brittleness.
Semiconducting polymers have previously been used as the transduction material in x-ray dosimeters, but these devices have a rather low detection sensitivity because of the low x-ray attenuation efficiency of the organic active layer. Here, we demonstrate a way to overcome this limitation through the introduction of high density nanoparticles having a high atomic number (Z) to increase the x-ray attenuation. Specifically, bismuth oxide (Bi O ) nanoparticles (Z=83 for Bi) are added to a poly(triarylamine) (PTAA) semiconducting polymer in the active layer of an x-ray detector. Scanning electron microscopy (SEM) reveals that the Bi O nanoparticles are reasonably distributed in the PTAA active layer. The reverse bias dc currentvoltage characteristics for PTAABi O diodes (with indium tin oxide (ITO) and Al contacts) have similar leakage currents to ITO/PTAA/Al diodes. Upon irradiation with 17.5keV x-ray beams, a PTAA device containing 60wt% Bi O nanoparticles demonstrates a sensitivity increase of approximately 2.5 times compared to the plain PTAA sensor. These results indicate that the addition of high-Z nanoparticles improves the performance of the dosimeters by increasing the x-ray stopping power of the active volume of the diode. Because the Bi O has a high density, it can be used very efficiently, achieving a high weight fraction with a low volume fraction of nanoparticles. The mechanical flexibility of the polymer is not sacrificed when the inorganic nanoparticles are incorporated. © 2012 IOP Publishing Ltd.
The non-equilibrium assembly of bimodal colloids during evaporative processes is an attractive means to achieve gradient or stratified layers in thick films. Here, we show that the stratification of small colloids on top of large is prevented when the viscosity of the continuous aqueous phase is too high. We propose a model where a too narrow width of the gradient in concentration of small colloids suppresses the stratification.
Polymer colloids are often copolymerized with acrylic acid monomers in order to impart colloidal stability. Here, the effects of the pH on the nanoscale and macroscopic adhesive properties of waterborne poly(butyl acrylate-co-acrylic acid) films are reported. In films cast from acidic colloidal dispersions, hydrogen bonding between carboxylic acid groups dominates the particle-particle interactions, whereas ionic dipolar interactions are dominant in films cast from basic dispersions. Force spectroscopy using an atomic force microscope and macroscale mechanical measurements show that latex films with hydrogen-bonding interactions have lower elastic moduli and are more deformable. They yield higher adhesion energies. On the other hand, in basic latex, ionic dipolar interactions increase the moduli of the dried films. These materials are stiffer and less deformable and, consequently, exhibit lower adhesion energies. The rate of water loss from acidic latex is slower, perhaps because of hydrogen bonding with the water. Therefore, although acid latex offers greater adhesion, there is a limitation in the film formation.
Flexible radiation dosimeters have been produced incorporating thick films (>1 μm) of the semiconducting polymer poly([9,9-dioctylfluorenyl-2,7-diyl]-co-bithiophene). Diode structures produced on aluminium-metallised poly(imide) substrates, and with gold top contacts, have been examined with respect to their electrical properties. The results suggest that a Schottky conduction mechanism occurs in the reverse biased diode, with a barrier to charge injection at the aluminium electrode. Optical absorption/emission spectra reveal a band gap of 2.48 eV for the polymer. The diodes have been used for direct charge detection of 17 keV X-rays, generated by a molybdenum source. Using operating voltages of -10 and -50 V respectively, sensitivities of 54 and 158 nC/mGy/cm3 have been achieved. Increasing the operating voltage shows that the diodes are stable up to approximately -200 V without significant increase in the dark current of the device (
The conventional method for synthesizing waterborne polymer colloids is emulsion polymerization using surfactants. An emerging method is the use of secondary dispersions (SD) of polymers in water, which avoids the addition of any surfactant. Although there are numerous studies of the water barrier properties (sorption, diffusion, and permeability) of waterborne emulsion (Em) polymer coatings, the properties of SD coatings, in comparison, have not been thoroughly investigated. Here, dynamic water vapor sorption analysis is used to compare the equilibrium sorption isotherms of the two forms of styrene-acrylate copolymers (Em and SD) with the same monomer composition. From an analysis of the kinetics of vapor sorption, the diffusion coefficient of water in the polymer coatings is determined. The combined effects of particle boundaries and surfactant addition were investigated through a comparison of the properties of SD and Em coatings to those of (1) solvent-cast polymer coatings (of the same monomer composition), (2) Em polymers that underwent dialysis to partially remove the water-soluble species, and (3) SD polymers with added surfactants. The results reveal that both the particle boundaries and the surfactants increase vapor sorption. The diffusion coefficients of water are comparable in magnitude in all of the polymer systems but are inversely related to water activity because of molecular clustering. Compared to all of the other waterborne polymer systems, the SD barrier coatings show the lowest equilibrium vapor sorption and permeability coefficients at high relative humidities as well as the lowest water diffusion coefficient at low humidities. These barrier properties make SD coatings an attractive alternative to conventional emulsion polymer coatings.
Using magnetic resonance profiling coupled with dynamic light scattering, we have investigated the mechanisms leading to the formation of a partly coalesced surface layer, or “open skin”, during film formation from waterborne polymer dispersions. We present the first use of the skewness of the distribution of free water as a model-free indicator of the spatial nonuniformity of drying. The skewness reaches a maximum at the same time at which a strong, static component, presumably originating from a skin at the film/air interface, appears in the light scattering data. Addition of salt to the dispersion increases both the skewness of the distribution of free water and the propensity for skin formation. Surprisingly, the drying is influenced not only by the concentration and valency of the ions in the salt but also by the particular ion. At intermediate particle densities, added salt strongly lowers the cooperative diffusion coefficient, Dcoop. When the particles reach close packing, Dcoop sharply increases. If the particles readily coalesce, the effects of the increased diffusivity will be counteracted, thereby inducing the formation of a skin. A modified Peclet number, Pe, using Dcoop, is proposed, so that the presence of salt is explicitly considered. This modified Pe is able to predict the nonuniformity in drying that leads to skin formation.
Many common elastomeric products, including nitrile gloves, are manufactured by coagulant dipping. This process involves the destabilization and gelation of a latex dispersion by an ionic coagulant. Despite widespread application, the physical chemistry governing coagulant dipping is poorly understood. It is unclear which properties of an electrolyte determine its efficacy as a coagulant and which phenomena control the growth of the gel. Here, a novel experimental protocol is developed to directly observe coagulant gelation by light microscopy. Gel growth is imaged and quantified for a variety of coagulants and compared to macroscopic dipping experiments mimicking the industrial process. When the coagulant is abundant, gels grow with a t1/2 time dependence, suggesting that this phenomenon is diffusion-dominated. When there is a finite amount of coagulant, gels grow to a limiting thickness. Both these situations are modeled as one-dimensional diffusion problems, reproducing the qualitative features of the experiments including which electrolytes cause rapid growth of thick gels. We propose that the gel thickness is limited by the amount of coagulant available, and the growth is, therefore, unbounded when the coagulant is abundant. The rate of the gel growth is controlled by a combination of a diffusion coefficient and the ratio of the critical coagulation concentration to the amount of coagulant present, which in many situations is set by the coagulant solubility. Other phenomena, including diffusiophoresis, may make a more minor contribution to the rate of gel growth.
Pressure-sensitive adhesives (PSAs) are usually made from viscoelastic, high-molecular weight copolymers, which are fine-tuned by adjusting the comonomer ratios, molecular weights, and cross-link densities to optimize the adhesion properties for the desired end-use. To create a lightly cross-linked network, an ultraviolet (UV) photoinitiator can be incorporated. Here, we present the first use of perfluorophenylazide chemistry to control precisely a polyacrylate network for application as a PSA. Upon UV irradiation, the highly reactive nitrene from the azide moiety reacts with nearby molecules through a C–H insertion reaction, resulting in cross-linking via covalent bonding. This approach offers three benefits: (1) a means to optimize adhesive properties without the addition of an external photoinitiator; (2) the ability to switch off the tack adhesion on demand via a high cross-linking density; and (3) a platform for additional chemical modification. A series of poly(n-butyl acrylate-co-2,3,4,5,6-pentafluorobenzyl acrylate) or poly(PFBA-co-BA) copolymers was synthesized and modified post-polymerization into the photo-reactive poly(n-butyl acrylate-co-4-azido-2,3,5,6-tetrafluorobenzyl acrylate) (azide-modified poly(PFBA-co-BA)) with various molar contents. When cast into films, the azide-modified copolymers with a high azide content achieved a very high shear resistance after UV irradiation, whereas the tack and peel adhesion decreased strongly with increasing azide content, indicating that excessive cross-linking occurred. These materials are thus photo-switchable. However, in the low range of azide content, an optimum probe tack adhesion energy was obtained in films with a 0.3 mol% azide content, where a long stress plateau (indicating good fibrillation) with a high plateau stress was observed. An optimum peel adhesion strength was achieved with 0.5 mol% azide. Thus, the adhesion was finely controlled by the degree of cross-linking of the PSA, as determined by the azide content of the copolymer chain. Finally, as a demonstration of the versatility and advantages of the materials platform, we show an azide–aldehyde–amine multicomponent modification of the azide copolymer to make a dye-functionalized film that retains its adhesive properties. This first demonstration of using azide functionality has enormous potential for functional PSA design.
A requirement for optimum performance in a pressure-sensitive adhesive (PSA) is the right balance between viscous and elastic properties, achieved by controlling the molecular architecture. Here, waterborne polyurethane PSAs are synthesized using a blend of polyether and polyalkene-based polyols. The effects of the polyol type and molecular weight on the adhesive and thermomechanical properties are explored to optimize them for PSA applications. A linear polyurethane is synthesized by the reaction of an aliphatic diisocyanate with a diol blend of polypropylene glycol (PPG) and hydroxyl-terminated polybutadiene (HTPB). With increasing concentrations of flexible HTPB segments and the associated increased viscous dissipation a favorable increase in the tack adhesion energy and peel strength is obtained. Adhesive properties are improved with increasing PPG molecular weight because chain entanglements become possible in the soft segments and raise the storage modulus. © 2013 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
The formation of solvent-cast, poly(methyl methacrylate) (PMMA) thin films from dilute bromobenzene solutions was studied using an ellipsometry technique. Bromobenzene has a relatively high refractive index (compared to PMMA), which provides contrast in ellipsometry, allowing the concentration to be determined. The solvent also has a relatively low evaporation rate, which makes the film formation slow enough to capture via the technique. The formation of the glassy film is thus studied in situ, and information on solvent and void concentration in the thin film during the film formation process is obtained. There is evidence that nanovoids (representing intramolecular space) develop in the film when solvent evaporates.
This chapter reviews the complex step of drying in the latex film formation process. Drying modes have a profound effect on drying rates and on the final properties of films, primarily through their influence on film morphology and the distribution of water-soluble species. Three distinct drying modes (acting separately, successively or together) can be defined, namely homogeneous drying (in which the water concentration remains uniform in the sample throughout the drying process), drying normal to the surface (where a dry layer of increasing thickness develops from the air surface of the latex coating); and lateral drying (where dry areas increase in size in a direction parallel to the substrate). Details are given on the current knowledge and understanding of these drying modes. The last section of the chapter considers the main parameters controlling the drying modes, i.e. thickness and geometric effects, the structure and rheology of the dispersion, particle viscoelasticity, and the overall rate of water loss.
Additional publications
A complete list of all of my publications can be seen here on my Google Scholar profile.